MASTER-NAADP: a membrane permeable precursor of the Ca2+ mobilizing second messenger NAADP
- PMID: 39271671
- PMCID: PMC11399135
- DOI: 10.1038/s41467-024-52024-y
MASTER-NAADP: a membrane permeable precursor of the Ca2+ mobilizing second messenger NAADP
Abstract
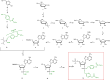
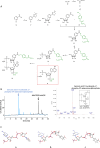
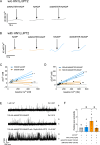
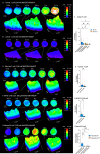
Upon stimulation of membrane receptors, nicotinic acid adenine dinucleotide phosphate (NAADP) is formed as second messenger within seconds and evokes Ca2+ signaling in many different cell types. Here, to directly stimulate NAADP signaling, MASTER-NAADP, a Membrane permeAble, STabilized, bio-rEversibly pRotected precursor of NAADP is synthesized and release of its active NAADP mimetic, benzoic acid C-nucleoside, 2'-phospho-3'F-adenosine-diphosphate, by esterase digestion is confirmed. In the presence of NAADP receptor HN1L/JPT2 (hematological and neurological expressed 1-like protein, HN1L, also known as Jupiter microtubule-associated homolog 2, JPT2), this active NAADP mimetic releases Ca2+ and increases the open probability of type 1 ryanodine receptor. When added to intact cells, MASTER-NAADP initially evokes single local Ca2+ signals of low amplitude. Subsequently, also global Ca2+ signaling is observed in T cells, natural killer cells, and Neuro2A cells. In contrast, control compound MASTER-NADP does not stimulate Ca2+ signaling. Likewise, in cells devoid of HN1L/JPT2, MASTER-NAADP does not affect Ca2+ signaling, confirming that the product released from MASTER-NAADP is a bona fide NAADP mimetic.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
- 335447717; SFB1328/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 236360313/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 516286863/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 236360313/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 516286863/Deutsche Forschungsgemeinschaft (German Research Foundation)
LinkOut - more resources
Full Text Sources
Miscellaneous

