Protein Biomarkers of Gastric Preneoplasia and Cancer Lesions in Blood: A Comprehensive Review
- PMID: 39272877
- PMCID: PMC11394471
- DOI: 10.3390/cancers16173019
Protein Biomarkers of Gastric Preneoplasia and Cancer Lesions in Blood: A Comprehensive Review
Abstract
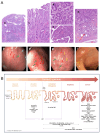
Gastric cancer (GC) is a major cause of cancer-related mortality worldwide. It is often associated with a bad prognosis because of its asymptomatic phenotype until advanced stages, highlighting the need for its prevention and early detection. GC development is preceded by the emergence of gastric preneoplasia lesions (GPNLs), namely atrophic gastritis (AG), intestinal metaplasia (IM), and dysplasia (DYS). GC is currently diagnosed by endoscopy, which is invasive and costly and has limited effectiveness for the detection of GPNLs. Therefore, the discovery of non-invasive biomarkers in liquid biopsies, such as blood samples, in order to identify the presence of gastric preneoplasia and/or cancer lesions at asymptomatic stages is of paramount interest. This comprehensive review provides an overview of recently identified plasma/serum proteins and their diagnostic performance for the prediction of GPNLs and early cancer lesions. Autoantibodies appear to be promising biomarkers for AG, IM and early gastric cancer detection, along with inflammation and immunity-related proteins and antibodies against H. pylori virulence factors. There is a lack of specific protein biomarkers with which to detect DYS. Despite the need for further investigation and validation, some emerging candidates could pave the way for the development of reliable, non-invasive diagnostic tests for the detection and prevention of GC.
Keywords: atrophic gastritis; diagnosis; dysplasia; early gastric cancer; intestinal metaplasia; liquid biopsy; plasma/serum; prevention; proteins.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Morgan E., Arnold M., Camargo M.C., Gini A., Kunzmann A.T., Matsuda T., Meheus F., Verhoeven R.H., Vignat J., Laversanne M., et al. The current and future incidence and mortality of gastric cancer in 185 countries, 2020–40: A population-based modelling study. eClinicalMedicine. 2022;47:101404. doi: 10.1016/j.eclinm.2022.101404. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

