The Expression of Toll-like Receptors in Cartilage Endplate Cells: A Role of Toll-like Receptor 2 in Pro-Inflammatory and Pro-Catabolic Gene Expression
- PMID: 39272974
- PMCID: PMC11394474
- DOI: 10.3390/cells13171402
The Expression of Toll-like Receptors in Cartilage Endplate Cells: A Role of Toll-like Receptor 2 in Pro-Inflammatory and Pro-Catabolic Gene Expression
Abstract
Introduction: The vertebral cartilage endplate (CEP), crucial for intervertebral disc health, is prone to degeneration linked to chronic low back pain, disc degeneration, and Modic changes (MC). While it is known that disc cells express toll-like receptors (TLRs) that recognize pathogen- and damage-associated molecular patterns (PAMPs and DAMPs), it is unclear if CEP cells (CEPCs) share this trait. The CEP has a higher cell density than the disc, making CEPCs an important contributor. This study aimed to identify TLRs on CEPCs and their role in pro-inflammatory and catabolic gene expression.
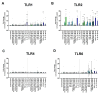
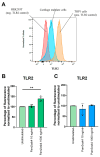
Methods: Gene expression of TLR1-10 was measured in human CEPs and expanded CEPCs using quantitative polymerase chain reaction. Additionally, surface TLR expression was measured in CEPs grouped into non-MC and MC. CEPCs were stimulated with tumor necrosis factor alpha, interleukin 1 beta, small-molecule TLR agonists, or the 30 kDa N-terminal fibronectin fragment. TLR2 signaling was inhibited with TL2-C29, and TLR2 protein expression was measured with flow cytometry.
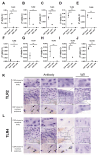
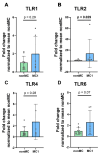
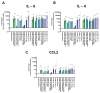
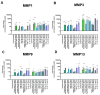
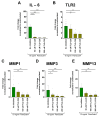
Results: Ex vivo analysis found all 10 TLRs expressed, while cultured CEPCs lost TLR8 and TLR9 expression. TLR2 expression was significantly increased in MC1 CEPCs, and its expression increased significantly after pro-inflammatory stimulation. Stimulation of the TLR2/6 heterodimer upregulated TLR2 protein expression. The TLR2/1 and TLR2/6 ligands upregulated pro-inflammatory genes and matrix metalloproteases (MMP1, MMP3, and MMP13), and TLR2 inhibition inhibited their upregulation. Endplate resorptive capacity of TLR2 activation was confirmed in a CEP explant model.
Conclusions: The expression of TLR1-10 in CEPCs suggests that the CEP is susceptible to PAMP and DAMP stimulation. Enhanced TLR2 expression in MC1, and generally in CEPCs under inflammatory conditions, has pro-inflammatory and pro-catabolic effects, suggesting a potential role in disc degeneration and MC.
Keywords: Modic changes; cartilage endplate; cartilage endplate cells; disc degeneration; toll-like receptors.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

References
-
- Wong J., Sampson S.L., Bell-Briones H., Ouyang A., Lazar A.A., Lotz J.C., Fields A. Nutrient supply and nucleus pulposus cell function: Effects of the transport properties of the cartilage endplate and potential implications for intradiscal biologic therapy. Osteoarthr. Cartil. 2019;27:956–964. doi: 10.1016/j.joca.2019.01.013. - DOI - PMC - PubMed
-
- Bailey J.F., Fields A.J., Ballatori A., Cohen D., Jain D., Coughlin D., O’neill C., McCormick Z., Han M., Krug R., et al. The Relationship Between Endplate Pathology and Patient-reported Symptoms for Chronic Low Back Pain Depends on Lumbar Paraspinal Muscle Quality. Spine (Phila Pa 1976) 2019;44:1010–1017. doi: 10.1097/BRS.0000000000003035. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

