Adipose-Derived Stem Cell Therapy in Spinal Cord Injury
- PMID: 39273075
- PMCID: PMC11394073
- DOI: 10.3390/cells13171505
Adipose-Derived Stem Cell Therapy in Spinal Cord Injury
Abstract
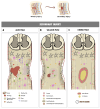
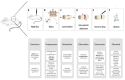
Spinal cord injury (SCI) is a serious condition accompanied by severe adverse events that affect several aspects of the patient's life, such as motor, sensory, and functional impairment. Despite its severe consequences, definitive treatment for these injuries is still missing. Therefore, researchers have focused on developing treatment strategies aimed at ensuring full recovery post-SCI. Accordingly, attention has been drawn toward cellular therapy using mesenchymal stem cells. Considering their wide availability, decreased immunogenicity, wide expansion capacity, and impressive effectiveness in many therapeutic approaches, adipose-derived stem cell (ADSC) injections in SCI cases have been investigated and showed promising results. In this review, SCI pathophysiology and ADSC transplantation benefits are discussed independently, together with SCI animal models and adipose stem cell preparation and application techniques. The mechanisms of healing in an SCI post-ADSC injection, the outcomes of this therapeutic approach, and current clinical trials are also deliberated, in addition to the challenges and future perspectives, aiming to encourage further research in this field.
Keywords: adipose tissue; spinal cord injury; stem cells; transplantation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Implantation of adipose-derived mesenchymal stem cell sheets promotes axonal regeneration and restores bladder function after spinal cord injury.Stem Cell Res Ther. 2022 Oct 12;13(1):503. doi: 10.1186/s13287-022-03188-1. Stem Cell Res Ther. 2022. PMID: 36224621 Free PMC article.
-
A combination of mesenchymal stem cells and scaffolds promotes motor functional recovery in spinal cord injury: a systematic review and meta-analysis.J Neurosurg Spine. 2019 Nov 1;32(2):269-284. doi: 10.3171/2019.8.SPINE19201. Print 2020 Feb 1. J Neurosurg Spine. 2019. PMID: 31675724
-
Intrathecal transplantation of autologous adipose-derived mesenchymal stem cells for treating spinal cord injury: A human trial.J Spinal Cord Med. 2016 Nov;39(6):655-664. doi: 10.1179/2045772315Y.0000000048. Epub 2015 Jul 24. J Spinal Cord Med. 2016. PMID: 26208177 Free PMC article. Clinical Trial.
-
Adipose-derived stem cell transplantation enhances spinal cord regeneration by upregulating PGRN expression.Neuroreport. 2024 Nov 6;35(16):1019-1029. doi: 10.1097/WNR.0000000000002091. Epub 2024 Sep 12. Neuroreport. 2024. PMID: 39292953
-
Human Mesenchymal Stem Cells for Spinal Cord Injury.Curr Stem Cell Res Ther. 2020;15(4):340-348. doi: 10.2174/1574888X15666200316164051. Curr Stem Cell Res Ther. 2020. PMID: 32178619 Review.
Cited by
-
Exploring NamiRNA networks and time-series gene expression in osteogenic differentiation of adipose-derived stem cells.Ann Med. 2025 Dec;57(1):2478323. doi: 10.1080/07853890.2025.2478323. Epub 2025 Mar 18. Ann Med. 2025. PMID: 40100054 Free PMC article.
References
-
- Spinal Cord Injury. [(accessed on 29 December 2023)]. Available online: https://www.who.int/news-room/fact-sheets/detail/spinal-cord-injury.
-
- Spinal Cord Injury—Symptoms and Causes. [(accessed on 29 December 2023)]. Available online: https://www.mayoclinic.org/diseases-conditions/spinal-cord-injury/sympto....
-
- GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators Global, Regional, and National Burden of Traumatic Brain Injury and Spinal Cord Injury, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:56–87. doi: 10.1016/S1474-4422(18)30415-0. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

