On the Stabilizing Effect of Aspartate and Glutamate and Its Counteraction by Common Denaturants
- PMID: 39273310
- PMCID: PMC11395698
- DOI: 10.3390/ijms25179360
On the Stabilizing Effect of Aspartate and Glutamate and Its Counteraction by Common Denaturants
Abstract
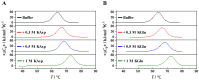
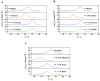
By performing differential scanning calorimetry(DSC) measurements on RNase A, we studied the stabilization provided by the addition of potassium aspartate(KAsp) or potassium glutamate (KGlu) and found that it leads to a significant increase in the denaturation temperature of the protein. The stabilization proves to be mainly entropic in origin. A counteraction of the stabilization provided by KAsp or KGlu is obtained by adding common denaturants such as urea, guanidinium chloride, or guanidinium thiocyanate. A rationalization of the experimental data is devised on the basis of a theoretical approach developed by one of the authors. The main contribution to the conformational stability of globular proteins comes from the gain in translational entropy of water and co-solute ions and/or molecules for the decrease in solvent-excluded volume associated with polypeptide folding (i.e., there is a large decrease in solvent-accessible surface area). The magnitude of this entropic contribution increases with the number density and volume packing density of the solution. The two destabilizing contributions come from the conformational entropy of the chain, which should not depend significantly on the presence of co-solutes, and from the direct energetic interactions between co-solutes and the protein surface in both the native and denatured states. It is the magnitude of the latter that discriminates between stabilizing and destabilizing agents.
Keywords: aspartate; counteraction; denaturants; glutamate; protein conformational stability.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Hofmeister F. Zur Lehre von der Wirkung der Salze: Zweite Mittheilung. Arch. Exp. Pathol. Pharmakol. 1888;24:247–260. doi: 10.1007/BF01918191. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

