Left Ventricular Systolic Dysfunction in NBCe1-B/C-Knockout Mice
- PMID: 39273556
- PMCID: PMC11395191
- DOI: 10.3390/ijms25179610
Left Ventricular Systolic Dysfunction in NBCe1-B/C-Knockout Mice
Abstract
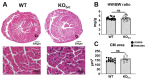
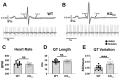
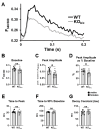
Congenital proximal renal tubular acidosis (pRTA) is a rare systemic disease caused by mutations in the SLC4A4 gene that encodes the electrogenic sodium bicarbonate cotransporter, NBCe1. The major NBCe1 protein variants are designated NBCe1-A, NBCe1-B, and NBCe1-C. NBCe1-A expression is kidney-specific, NBCe1-B is broadly expressed and is the only NBCe1 variant expressed in the heart, and NBCe1-C is a splice variant of NBCe1-B that is expressed in the brain. No cardiac manifestations have been reported from patients with pRTA, but studies in adult rats with virally induced reduction in cardiac NBCe1-B expression indicate that NBCe1-B loss leads to cardiac hypertrophy and prolonged QT intervals in rodents. NBCe1-null mice die shortly after weaning, so the consequence of congenital, global NBCe1 loss on the heart is unknown. To circumvent this issue, we characterized the cardiac function of NBCe1-B/C-null (KOb/c) mice that survive up to 2 months of age and which, due to the uninterrupted expression of NBCe1-A, do not exhibit the confounding acidemia of the globally null mice. In contrast to the viral knockdown model, cardiac hypertrophy was not present in KOb/c mice as assessed by heart-weight-to-body-weight ratios and cardiomyocyte cross-sectional area. However, echocardiographic analysis revealed reduced left ventricular ejection fraction, and intraventricular pressure-volume measurements demonstrated reduced load-independent contractility. We also observed increased QT length variation in KOb/c mice. Finally, using the calcium indicator Fura-2 AM, we observed a significant reduction in the amplitude of Ca2+ transients in paced KOb/c cardiomyocytes. These data indicate that congenital, global absence of NBCe1-B/C leads to impaired cardiac contractility and increased QT length variation in juvenile mice. It remains to be determined whether the cardiac phenotype in KOb/c mice is influenced by the absence of NBCe1-B/C from neuronal and endocrine tissues.
Keywords: NBCe1; acid–base; calcium; contractility; heart failure; pRTA.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
-
- Horita S., Simsek E., Simsek T., Yildirim N., Ishiura H., Nakamura M., Satoh N., Suzuki A., Tsukada H., Mizuno T., et al. SLC4A4 Compound Heterozygous Mutations in Exon–Intron Boundary Regions Presenting with Severe Proximal Renal Tubular Acidosis and Extrarenal Symptoms Coexisting with Turner’s Syndrome: A Case Report. BMC Med. Genet. 2018;19:103. doi: 10.1186/s12881-018-0612-y. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

