MeCP2 is a naturally supercharged protein with cell membrane transduction capabilities
- PMID: 39276009
- PMCID: PMC11400631
- DOI: 10.1002/pro.5170
MeCP2 is a naturally supercharged protein with cell membrane transduction capabilities
Abstract
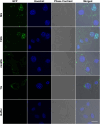
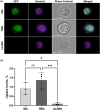
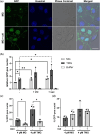
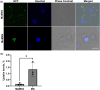
The intrinsically disordered protein MeCP2 is a global transcriptional regulator encoded by the MECP2 gene. Although the structured domains of MeCP2 have been the subject of multiple studies, its unstructured regions have not been that extensively characterized. In this work, we show that MeCP2 possesses properties akin to those of supercharged proteins. By utilizing its unstructured portions, MeCP2 can successfully transduce across cell membranes and localize to heterochromatic foci in the nuclei, displaying uptake levels a third lower than a MeCP2 construct fused to the cell-penetrating peptide TAT. MeCP2 uptake can further be enhanced by the addition of compounds that promote endosomal escape following cellular trafficking by means of macropinocytosis. Using a combination of in silico prediction algorithms and live-cell imaging experiments, we mapped the sequence in MeCP2 responsible for its cellular incorporation, which bears a striking resemblance to TAT itself. Transduced MeCP2 was shown to interact with HDAC3. These findings provide valuable insight into the properties of MeCP2 and may be beneficial for devising future protein-based treatment strategies.
Keywords: MeCP2; TAT fusion proteins; cell penetrating peptides; supercharged protein.
© 2024 The Author(s). Protein Science published by Wiley Periodicals LLC on behalf of The Protein Society.
Conflict of interest statement
The authors A. V. B., A. H., V. S., A. S., N. S., and T. S. declare no conflicts of interest.
Figures


Similar articles
-
Expression, Purification, Characterization and Cellular Uptake of MeCP2 Variants.Protein J. 2022 Apr;41(2):345-359. doi: 10.1007/s10930-022-10054-9. Epub 2022 May 12. Protein J. 2022. PMID: 35546650 Free PMC article.
-
Influence of the disordered domain structure of MeCP2 on its structural stability and dsDNA interaction.Int J Biol Macromol. 2021 Apr 1;175:58-66. doi: 10.1016/j.ijbiomac.2021.01.206. Epub 2021 Feb 3. Int J Biol Macromol. 2021. PMID: 33548325
-
Intrinsic disorder and autonomous domain function in the multifunctional nuclear protein, MeCP2.J Biol Chem. 2007 May 18;282(20):15057-64. doi: 10.1074/jbc.M700855200. Epub 2007 Mar 19. J Biol Chem. 2007. PMID: 17371874
-
Progress in Research and Application of HIV-1 TAT-Derived Cell-Penetrating Peptide.J Membr Biol. 2017 Apr;250(2):115-122. doi: 10.1007/s00232-016-9940-z. Epub 2016 Dec 8. J Membr Biol. 2017. PMID: 27933338 Review.
-
Regulation, diversity and function of MECP2 exon and 3'UTR isoforms.Hum Mol Genet. 2020 Sep 30;29(R1):R89-R99. doi: 10.1093/hmg/ddaa154. Hum Mol Genet. 2020. PMID: 32681172 Free PMC article. Review.
References
-
- Albarran B, To R, Stayton PS. A TAT–streptavidin fusion protein directs uptake of biotinylated cargo into mammalian cells. Protein Eng Des Sel. 2005;18(3):147–152. - PubMed
-
- Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY. Rett syndrome is caused by mutations in x‐linked MECP2, encoding methyl‐cpg‐binding protein 2. Nat Genet. 1999;23(2):185–188. - PubMed
-
- Buschdorf JP, Stratling WH. A WW domain binding region in methyl‐cpg‐binding protein MeCP2: impact on Rett syndrome. J Mol Med (Berl). 2004;82(2):135–143. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

