Engineered FHA domains can bind to a variety of Phosphothreonine-containing peptides
- PMID: 39276365
- PMCID: PMC11436287
- DOI: 10.1093/protein/gzae014
Engineered FHA domains can bind to a variety of Phosphothreonine-containing peptides
Abstract
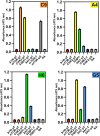
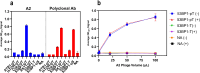
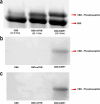
Antibodies play a crucial role in monitoring post-translational modifications, like phosphorylation, which regulates protein activity and location; however, commercial polyclonal and monoclonal antibodies have limitations in renewability and engineering compared to recombinant affinity reagents. A scaffold based on the Forkhead-associated domain (FHA) has potential as a selective affinity reagent for this post-translational modification. Engineered FHA domains, termed phosphothreonine-binding domains (pTBDs), with limited cross-reactivity were isolated from an M13 bacteriophage display library by affinity selection with phosphopeptides corresponding to human mTOR, Chk2, 53BP1, and Akt1 proteins. To determine the specificity of the representative pTBDs, we focused on binders to the pT543 phosphopeptide (536-IDEDGENpTQIEDTEP-551) of the DNA repair protein 53BP1. ELISA and western blot experiments have demonstrated the pTBDs are specific to phosphothreonine, demonstrating the potential utility of pTBDs for monitoring the phosphorylation of specific threonine residues in clinically relevant human proteins.
Keywords: 53BP1; Akt1; Chk2; FHA domain; affinity selection; library; mTOR; phosphoepitope; phosphopeptide; phosphoserine; phosphothreonine; sortase.
© The Author(s) 2024. Published by Oxford University Press. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

