Corilagin alleviates podocyte injury in diabetic nephropathy by regulating autophagy via the SIRT1-AMPK pathway
- PMID: 39280180
- PMCID: PMC11372637
- DOI: 10.4239/wjd.v15.i9.1916
Corilagin alleviates podocyte injury in diabetic nephropathy by regulating autophagy via the SIRT1-AMPK pathway
Abstract
Background: Diabetic nephropathy (DN) is the most frequent chronic microvascular consequence of diabetes, and podocyte injury and malfunction are closely related to the development of DN. Studies have shown that corilagin (Cor) has hepatoprotective, anti-inflammatory, antibacterial, antioxidant, anti-hypertensive, anti-diabetic, and anti-tumor activities.
Aim: To explore the protective effect of Cor against podocyte injury in DN mice and the underlying mechanisms.
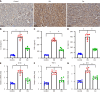
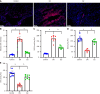
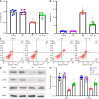
Methods: Streptozotocin and a high-fat diet were combined to generate DN mice models, which were then divided into either a Cor group or a DN group (n = 8 in each group). Mice in the Cor group were intraperitoneally injected with Cor (30 mg/kg/d) for 12 wk, and mice in the DN group were treated with saline. Biochemical analysis was used to measure the blood lipid profiles. Hematoxylin and eosin staining was used to detect pathological changes in kidney tissue. Immunohistochemistry and Western blotting were used to assess the protein expression of nephrin and podocin. Mouse podocyte cells (MPC5) were cultured and treated with glucose (5 mmol/L), Cor (50 μM), high glucose (HG) (30 mmol/L), and HG (30 mmol/L) plus Cor (50 μM). Real-time quantitative PCR and Western blotting were performed to examine the effects of Cor on podocyte autophagy.
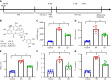
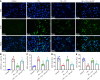
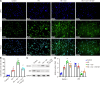
Results: Compared with the control group, the DN mice models had increased fasting blood glucose, glycosylated hemoglobin, triglycerides, and total cholesterol, decreased nephrin and podocin expression, increased apoptosis rate, elevated inflammatory cytokines, and enhanced oxidative stress. All of the conditions mentioned above were alleviated after intervention with Cor. In addition, Cor therapy improved SIRT1 and AMPK expression (P < 0.001), inhibited reactive oxygen species and oxidative stress, and elevated autophagy in HG-induced podocytes (P < 0.01).
Conclusion: Cor alleviates podocyte injury by regulating autophagy via the SIRT1-AMPK pathway, thereby exerting its protective impact on renal function in DN mice.
Keywords: Autophagy; Corilagin; Diabetic nephropathy; High glucose; Podocyte injury; SIRT1-AMPK pathway.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



References
LinkOut - more resources
Full Text Sources

