Antibodies as key mediators of protection against Mycobacterium tuberculosis
- PMID: 39286260
- PMCID: PMC11402706
- DOI: 10.3389/fimmu.2024.1430955
Antibodies as key mediators of protection against Mycobacterium tuberculosis
Abstract
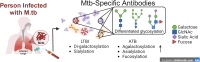
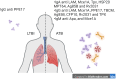
Tuberculosis (TB) is caused by infection with the bacterial pathogen Mycobacterium tuberculosis (M.tb) in the respiratory tract. There was an estimated 10.6 million people newly diagnosed with TB, and there were approximately 1.3 million deaths caused by TB in 2022. Although the global prevalence of TB has remained high for decades and is an annual leading cause of death attributed to infectious diseases, only one vaccine, Bacillus Calmette-Guérin (BCG), has been approved so far to prevent/attenuate TB disease. Correlates of protection or immunological mechanisms that are needed to control M.tb remain unknown. The protective role of antibodies after BCG vaccination has also remained largely unclear; however, recent studies have provided evidence for their involvement in protection against disease, as biomarkers for the state of infection, and as potential predictors of outcomes. Interestingly, the antibodies generated post-vaccination with BCG are linked to the activation of innate immune cascades, providing further evidence that antibody effector functions are critical for protection against respiratory pathogens such as M.tb. In this review, we aim to provide current knowledge of antibody application in TB diagnosis, prevention, and treatment. Particularly, this review will focus on 1) The role of antibodies in preventing M.tb infections through preventing Mtb adherence to epithelium, antibody-mediated phagocytosis, and antibody-mediated cellular cytotoxicity; 2) The M.tb-directed antibody response generated after vaccination and how humoral profiles with different glycosylation patterns of these antibodies are linked with protection against the disease state; and 3) How antibody-mediated immunity against M.tb can be further explored as early diagnosis biomarkers and different detection methods to combat the global M.tb burden. Broadening the paradigm of differentiated antibody profiling and antibody-based detection during TB disease progression offers new directions for diagnosis, treatment, and preventative strategies. This approach involves linking the aforementioned humoral responses with the disease state, progression, and clearance.
Keywords: LTBI (latent TB infection); active tuberculosis (ATB); antibody; biomarkers; serology; systems immunology; tuberculosis.
Copyright © 2024 Wang, Nag, Baldwin, Coler and McNamara.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

References
-
- WHO . Global tuberculosis report 2023. World Health Organization (WHO) (2023).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

