Mathematical Models of Ocular Drug Delivery
- PMID: 39287588
- PMCID: PMC11412384
- DOI: 10.1167/iovs.65.11.28
Mathematical Models of Ocular Drug Delivery
Abstract
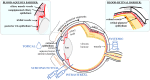
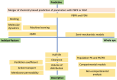
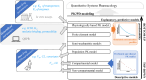
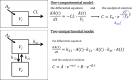
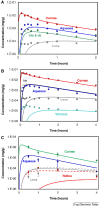
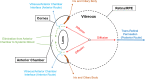
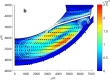
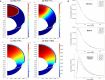
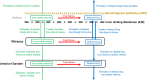
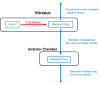
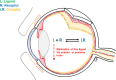
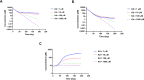
Drug delivery is an important factor for the success of ocular drug treatment. However, several physical, biochemical, and flow-related barriers limit drug exposure of anterior and posterior ocular target tissues during drug treatment via topical, subconjunctival, intravitreal, or systemic routes. Mathematical models encompass various barriers so that their joint influence on pharmacokinetics (PKs) can be simulated in an integrated fashion. The models are useful in predicting PKs and even pharmacodynamics (PDs) of administered drugs thereby fostering development of new drug molecules and drug delivery systems. Furthermore, the models are potentially useful in interspecies translation and probing of disease effects on PKs. In this review article, we introduce current modeling methods (noncompartmental analyses, compartmental and physiologically based PK models, and finite element models) in ocular PKs and related drug delivery. The roles of top-down models and bottom-up simulations are discussed. Furthermore, we present some future challenges, such as modeling of intra-tissue distribution, prediction of drug responses, quantitative systems pharmacology, and possibilities of artificial intelligence.
Conflict of interest statement
Disclosure:
Figures

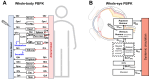

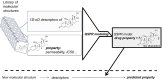
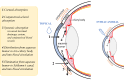
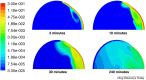


References
-
- Urtti A. Challenges and obstacles of ocular pharmacokinetics and drug delivery. Adv Drug Deliv Rev. 2006; 58(11): 1131–1135. - PubMed
-
- Fayyaz A, Ranta VP, Toropainen E, et al.. Topical ocular pharmacokinetics and bioavailability for a cocktail of atenolol, timolol and betaxolol in rabbits. Eur J Pharm Sci. 2020; 155: 105553. - PubMed
-
- del Amo EM, Urtti A.. Current and future ophthalmic drug delivery systems. A shift to the posterior segment. Drug Discov Today. 2008; 13(3): 135–143. - PubMed
-
- Del Amo EM, Vellonen KS, Kidron H, Urtti A.. Intravitreal clearance and volume of distribution of compounds in rabbits: in silico prediction and pharmacokinetic simulations for drug development. Eur Journal of Pharm Biopharm. 2015; 95: 215–226. - PubMed
-
- Zhu Q, Ziemssen F, Henke-Fahle S, et al.. Vitreous levels of bevacizumab and vascular endothelial growth factor-A in patients with choroidal neovascularization. Ophthalmology. 2008; 115(10): 1750–1755.e1. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

