mLumiOpto Is a Mitochondrial-Targeted Gene Therapy for Treating Cancer
- PMID: 39288077
- PMCID: PMC11609628
- DOI: 10.1158/0008-5472.CAN-24-0984
mLumiOpto Is a Mitochondrial-Targeted Gene Therapy for Treating Cancer
Abstract
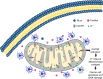
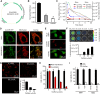
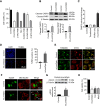
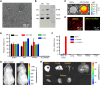
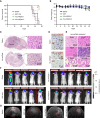
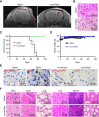
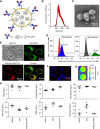
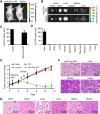
Mitochondria are important in various aspects of cancer development and progression. Targeting mitochondria in cancer cells holds great therapeutic promise, yet current strategies to specifically and effectively destroy cancer mitochondria in vivo are limited. Here, we developed mitochondrial luminoptogenetics (mLumiOpto), an innovative mitochondrial-targeted luminoptogenetics gene therapy designed to directly disrupt the inner mitochondrial membrane potential and induce cancer cell death. The therapeutic approach included synthesis of a blue light-gated cationic channelrhodopsin in the inner mitochondrial membrane and coexpression of a blue bioluminescence-emitting nanoluciferase in the cytosol of the same cells. The mLumiOpto genes were selectively delivered to cancer cells in vivo by an adeno-associated virus carrying a cancer-specific promoter or cancer-targeted mAB-tagged exosome-associated adeno-associated virus. Induction with nanoluciferase luciferin elicited robust endogenous bioluminescence, which activated cationic channelrhodopsin, triggering cancer cell mitochondrial depolarization and subsequent cell death. Importantly, mLumiOpto demonstrated remarkable efficacy in reducing tumor burden and killing tumor cells in glioblastoma and triple-negative breast cancer xenograft mouse models. Furthermore, the approach induced an antitumor immune response, increasing infiltration of dendritic cells and CD8+ T cells in the tumor microenvironment. These findings establish mLumiOpto as a promising therapeutic strategy by targeting cancer cell mitochondria in vivo. Significance: mLumiOpto is a next generation optogenetic approach that employs selective delivery of genes to cancer cells to trigger mitochondrial depolarization, effectively inducing cell death and reducing tumor burden.
©2024 The Authors; Published by the American Association for Cancer Research.
Conflict of interest statement
No disclosures were reported.
Figures
Update of
-
An Innovative Mitochondrial-targeted Gene Therapy for Cancer Treatment.bioRxiv [Preprint]. 2024 Mar 27:2024.03.24.584499. doi: 10.1101/2024.03.24.584499. bioRxiv. 2024. Update in: Cancer Res. 2024 Dec 2;84(23):4049-4065. doi: 10.1158/0008-5472.CAN-24-0984. PMID: 38585739 Free PMC article. Updated. Preprint.
References
-
- Stanley WC, Recchia FA, Lopaschuk GD. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev 2005;85:1093–129. - PubMed
-
- Neuzil J, Wang X-F, Dong L-F, Low P, Ralph SJ. Molecular mechanism of ‘mitocan’-induced apoptosis in cancer cells epitomizes the multiple roles of reactive oxygen species and Bcl-2 family proteins. FEBS Lett 2006;580:5125–9. - PubMed
-
- Horobin RW, Trapp S, Weissig V. Mitochondriotropics: a review of their mode of action, and their applications for drug and DNA delivery to mammalian mitochondria. J Control Release 2007;121:125–36. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

