Mouse polyomavirus infection induces lamin reorganisation
- PMID: 39288210
- PMCID: PMC11616003
- DOI: 10.1111/febs.17275
Mouse polyomavirus infection induces lamin reorganisation
Abstract
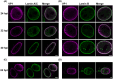
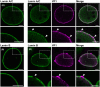
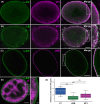
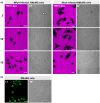
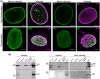
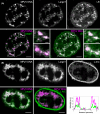
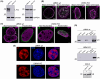
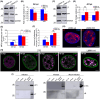
The nuclear lamina is a dense network of intermediate filaments beneath the inner nuclear membrane. Composed of A-type lamins (lamin A/C) and B-type lamins (lamins B1 and B2), the nuclear lamina provides a scaffold for the nuclear envelope and chromatin, thereby maintaining the structural integrity of the nucleus. A-type lamins are also found inside the nucleus where they interact with chromatin and participate in gene regulation. Viruses replicating in the cell nucleus have to overcome the nuclear envelope during the initial phase of infection and during the nuclear egress of viral progeny. Here, we focused on the role of lamins in the replication cycle of a dsDNA virus, mouse polyomavirus. We detected accumulation of the major capsid protein VP1 at the nuclear periphery, defects in nuclear lamina staining and different lamin A/C phosphorylation patterns in the late phase of mouse polyomavirus infection, but the nuclear envelope remained intact. An absence of lamin A/C did not affect the formation of replication complexes but did slow virus propagation. Based on our findings, we propose that the nuclear lamina is a scaffold for replication complex formation and that lamin A/C has a crucial role in the early phases of infection with mouse polyomavirus.
Keywords: VP1; lamin A/C; lamin B; mouse polyomavirus; viral replication centres.
© 2024 The Author(s). The FEBS Journal published by John Wiley & Sons Ltd on behalf of Federation of European Biochemical Societies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Nuclear envelope remodelling during human spermiogenesis involves somatic B-type lamins and a spermatid-specific B3 lamin isoform.Mol Hum Reprod. 2015 Mar;21(3):225-36. doi: 10.1093/molehr/gau111. Epub 2014 Dec 4. Mol Hum Reprod. 2015. PMID: 25477337
-
Lamins: The backbone of the nucleocytoskeleton interface.Curr Opin Cell Biol. 2024 Feb;86:102313. doi: 10.1016/j.ceb.2023.102313. Epub 2024 Jan 22. Curr Opin Cell Biol. 2024. PMID: 38262116 Review.
-
Nuclear pore protein TPR associates with lamin B1 and affects nuclear lamina organization and nuclear pore distribution.Cell Mol Life Sci. 2019 Jun;76(11):2199-2216. doi: 10.1007/s00018-019-03037-0. Epub 2019 Feb 14. Cell Mol Life Sci. 2019. PMID: 30762072 Free PMC article.
-
Laminopathies: what can humans learn from fruit flies.Cell Mol Biol Lett. 2018 Jul 6;23:32. doi: 10.1186/s11658-018-0093-1. eCollection 2018. Cell Mol Biol Lett. 2018. PMID: 30002683 Free PMC article. Review.
-
Partial cleavage of A-type lamins concurs with their total disintegration from the nuclear lamina during apoptosis.Eur J Cell Biol. 2002 Dec;81(12):677-91. doi: 10.1078/0171-9335-00282. Eur J Cell Biol. 2002. PMID: 12553668
Cited by
-
Intermediate Filament Protein BFSP1 Maintains Oocyte Asymmetric Division by Modulating Spindle Length.Adv Sci (Weinh). 2025 Jul;12(28):e2504066. doi: 10.1002/advs.202504066. Epub 2025 May 11. Adv Sci (Weinh). 2025. PMID: 40349178 Free PMC article.
References
-
- Aebi U, Cohn J, Buhle L & Gerace L (1986) The nuclear lamina is a meshwork of intermediate‐type filaments. Nature 323, 560–564. - PubMed
-
- Gerace L & Burke B (1988) Functional organization of the nuclear envelope. Annu Rev Cell Biol 4, 335–374. - PubMed
-
- Nmezi B, Xu J, Fu R, Armiger TJ, Rodriguez‐Bey G, Powell JS, Ma H, Sullivan M, Tu Y, Chen NY et al. (2019) Concentric organization of A‐ and B‐type lamins predicts their distinct roles in the spatial organization and stability of the nuclear lamina. Proc Natl Acad Sci USA 116, 4307–4315. - PMC - PubMed
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

