COBRA N2 NA vaccines induce protective immune responses against influenza viral infection
- PMID: 39291424
- PMCID: PMC11657087
- DOI: 10.1080/21645515.2024.2403175
COBRA N2 NA vaccines induce protective immune responses against influenza viral infection
Abstract
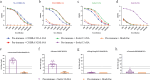
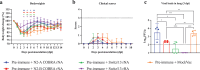
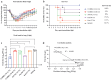
Influenza neuraminidase (NA) is a promising target for a broadly protective vaccine. In this study, the Computationally Optimized Broadly Reactive Antigen (COBRA) methodology was used to develop N2 NA vaccine candidates. The unique wild type (WT) N2 sequences of human and swine influenza strains isolated between 1957 and 2019 were used to design the COBRA N2-A NA vaccine, while the unique WT N2 sequences of human influenza strains isolated between 2000 and 2019 were used to design the COBRA N2-B NA vaccine. Sera collected from COBRA N2 NA vaccinated mice showed more broadly reactive antibody responses against a broad panel of H×N2 influenza virus strains than sera collected from mice vaccinated with WT N2 NA vaccines. Antibodies elicited by COBRA or WT N2 NA antigens cross react with recent human H3N2 influenza viruses from different clades, while the antibodies elicited by A/Switzerland/9715293/2013 hemagglutinin (HA) reacted with viruses from the same clade. Furthermore, mice vaccinated with COBRA N2-B NA vaccine had lower viral lung titers compared to mock vaccinated mice when challenged with human H3N2 influenza viruses. Thus, the COBRA N2 NA vaccines elicit broadly protective murine anti-NA antibodies against multiple strains across subtypes and the viral loads were significantly decreased in the lungs of the mice in the COBRA N2 NA vaccine groups, compared to the mice in the mock vaccinated group, indicating that the COBRA-based N2 subtype NA vaccines have a potential to be a component in a universal influenza vaccine.
Keywords: COBRA; N2; influenza; mice; neuraminidase (NA); vaccine.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures



Similar articles
-
Universal Influenza Virus Neuraminidase Vaccine Elicits Protective Immune Responses against Human Seasonal and Pre-pandemic Strains.J Virol. 2021 Aug 10;95(17):e0075921. doi: 10.1128/JVI.00759-21. Epub 2021 Aug 10. J Virol. 2021. PMID: 34160258 Free PMC article.
-
Computationally Optimized Broadly Reactive Hemagglutinin Elicits Hemagglutination Inhibition Antibodies against a Panel of H3N2 Influenza Virus Cocirculating Variants.J Virol. 2017 Nov 30;91(24):e01581-17. doi: 10.1128/JVI.01581-17. Print 2017 Dec 15. J Virol. 2017. PMID: 28978710 Free PMC article.
-
Elicitation of Protective Antibodies against 20 Years of Future H3N2 Cocirculating Influenza Virus Variants in Ferrets Preimmune to Historical H3N2 Influenza Viruses.J Virol. 2019 Jan 17;93(3):e00946-18. doi: 10.1128/JVI.00946-18. Print 2019 Feb 1. J Virol. 2019. PMID: 30429350 Free PMC article.
-
Anti-neuraminidase immunity in the combat against influenza.Expert Rev Vaccines. 2024 Jan-Dec;23(1):474-484. doi: 10.1080/14760584.2024.2343689. Epub 2024 Apr 23. Expert Rev Vaccines. 2024. PMID: 38632930 Free PMC article. Review.
-
Neuraminidase as an influenza vaccine antigen: a low hanging fruit, ready for picking to improve vaccine effectiveness.Curr Opin Immunol. 2018 Aug;53:38-44. doi: 10.1016/j.coi.2018.03.025. Epub 2018 Apr 16. Curr Opin Immunol. 2018. PMID: 29674167 Free PMC article. Review.
Cited by
-
Multivalent H3 COBRA-based influenza vaccine elicits enhanced immune response in a pre-immune elderly ferret model.Vaccine. 2025 May 22;56:127156. doi: 10.1016/j.vaccine.2025.127156. Epub 2025 Apr 22. Vaccine. 2025. PMID: 40267617
References
-
- Iuliano AD, Roguski KM, Chang HH, Muscatello DJ, Palekar R, Tempia S, Cohen C, Gran JM, Schanzer D, Cowling BJ, et al. Estimates of global seasonal influenza-associated respiratory mortality: a modelling study. The Lancet. 2018;391(10127):1285–12. doi:10.1016/S0140-6736(17)33293-2. - DOI - PMC - PubMed
-
- Sandbulte MR, Westgeest KB, Gao J, Xu X, Klimov AI, Russell CA, Burke DF, Smith DJ, Fouchier RAM, Eichelberger MC, et al. Discordant antigenic drift of neuraminidase and hemagglutinin in H1N1 and H3N2 influenza viruses. Proc Natl Acad Sci USA. 2011;108(51):20748–53. doi:10.1073/pnas.1113801108. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
