A study protocol for a randomized clinical trial on exposure and effects of pesticides consumption - the PEST-EXPO Brazil study
- PMID: 39295633
- PMCID: PMC11408870
- DOI: 10.1016/j.mex.2024.102942
A study protocol for a randomized clinical trial on exposure and effects of pesticides consumption - the PEST-EXPO Brazil study
Abstract
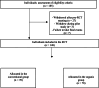
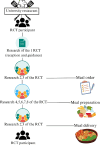
Randomized clinical trials are considered the gold standard for studies with dietary interventions, which is mainly due to the fact that they can establish causal relationships between food exposure and body composition measures or biomarkers. The aim of this study was to describe the details of a double-blind, randomized, clinical trial protocol to identify, characterize and evaluate the effects of human dietary exposure to pesticide residues in food. Specific aspects of planning (development of a research question, determination of objectives, selection of participants, randomization and blinding) and performance (recruitment of participants, measures to improve adherence, data collection, follow-up and evaluation of results) are addressed in this study. The study design proved effective in characterizing dietary patterns with foods originating from both conventional and organic agriculture. A total of 148 individuals were recruited for the study. The conventional group was represented by 47 % of the sample and the organic group was represented by 53 %. The practice of evidence-based nutrition has demanded that trials be well designed and systematically performed in the field of clinical nutrition. Therefore, this clinical trial emphasizes the importance of improving studies with toxicological nutrition that assess sources of exposure through food.•This double-blind, randomized clinical trial details the protocol for identifying, characterizing, and evaluating the effects of dietary exposure to pesticide residues.•The protocol demonstrates that well-designed and systematically conducted trials emphasize the importance of robust methodologies in evidence-based nutrition.•In the face of the global climate crisis, this clinical trial underscores the importance of enhancing studies in toxicological nutrition, particularly those evaluating sources of exposure through food, to better understand the dietary impacts on health.
Keywords: Nutrition; PEST-EXPO Brazil study; Pesticide; Toxicology; Trial methodology.
© 2024 The Authors. Published by Elsevier B.V.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
References
-
- Fortes C., Mastroeni S., Pilla M.A., Antonelli G., Lunghini L., Aprea C. The relation between dietary habits and urinary levels of 3-phenoxybenzoic acid, a pyrethroid metabolite. Food Chem. Toxicol. 2013;2:91–96. - PubMed
-
- Xue J., Zartarian V., Tornero-Velez R., Tulve N.S. EPA's SHEDS-multimedia model: children's cumulative pyrethroid exposure estimates and evaluation against NHANES biomarker data. Environ. Int. 2014;73:304–311. - PubMed
LinkOut - more resources
Full Text Sources