Novel systematic processing of cardiac magnetic resonance imaging identifies target regions associated with infarct-related ventricular tachycardia
- PMID: 39298664
- PMCID: PMC11472157
- DOI: 10.1093/europace/euae244
Novel systematic processing of cardiac magnetic resonance imaging identifies target regions associated with infarct-related ventricular tachycardia
Abstract
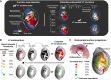
Aims: There is lack of agreement on late gadolinium enhancement cardiac magnetic resonance (LGE-CMR) imaging processing for guiding ventricular tachycardia (VT) ablation. We aim at developing and validating a systematic processing approach on LGE-CMR images to identify VT corridors that contain critical VT isthmus sites.
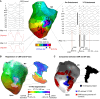
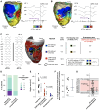
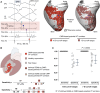
Methods and results: This is a translational study including 18 pigs with established myocardial infarction and inducible VT undergoing in vivo characterization of the anatomical and functional myocardial substrate associated with VT maintenance. Clinical validation was conducted in a multicentre series of 33 patients with ischaemic cardiomyopathy undergoing VT ablation. Three-dimensional LGE-CMR images were processed using systematic scanning of 15 signal intensity (SI) cut-off ranges to obtain surface visualization of all potential VT corridors. Analysis and comparisons of imaging and electrophysiological data were performed in individuals with full electrophysiological characterization of the isthmus sites of at least one VT morphology. In both the experimental pig model and patients undergoing VT ablation, all the electrophysiologically defined isthmus sites (n = 11 and n = 19, respectively) showed overlapping regions with CMR-based potential VT corridors. Such imaging-based VT corridors were less specific than electrophysiologically guided ablation lesions at critical isthmus sites. However, an optimized strategy using the 7 most relevant SI cut-off ranges among patients showed an increase in specificity compared to using 15 SI cut-off ranges (70 vs. 62%, respectively), without diminishing the capability to detect VT isthmus sites (sensitivity 100%).
Conclusion: Systematic imaging processing of LGE-CMR sequences using several SI cut-off ranges may improve and standardize procedure planning to identify VT isthmus sites.
Keywords: Imaging processing; Magnetic resonance imaging; Radiofrequency ablation; Ventricular tachycardia.
© The Author(s) 2024. Published by Oxford University Press on behalf of the European Society of Cardiology.
Conflict of interest statement
Conflict of interest: none declared.
Figures



References
-
- Tung R, Raiman M, Liao H, Zhan X, Chung FP, Nagel R et al. Simultaneous endocardial and epicardial delineation of 3D reentrant ventricular tachycardia. J Am Coll Cardiol 2020;75:884–97. - PubMed
-
- Nishimura T, Shatz N, Weiss P, Zawaneh M, Bai R, Beaser AD et al. Identification of human ventricular tachycardia demarcated by fixed lines of conduction block in a 3-dimensional hyperboloid circuit. Circulation 2023;148:1354–67. - PubMed
-
- de Chillou C, Groben L, Magnin-Poull I, Andronache M, MagdiAbbas M, Zhang N et al. Localizing the critical isthmus of postinfarct ventricular tachycardia: the value of pace-mapping during sinus rhythm. Heart Rhythm 2014;11:175–81. - PubMed
-
- Di Biase L, Santangeli P, Burkhardt DJ, Bai R, Mohanty P, Carbucicchio C et al. Endo-epicardial homogenization of the scar versus limited substrate ablation for the treatment of electrical storms in patients with ischemic cardiomyopathy. J Am Coll Cardiol 2012;60:132–41. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

