Disproportionality Analysis and Characterisation of Medication Errors in EudraVigilance: Exploring Findings on Sexes and Age Groups
- PMID: 39300043
- PMCID: PMC11711134
- DOI: 10.1007/s40264-024-01478-6
Disproportionality Analysis and Characterisation of Medication Errors in EudraVigilance: Exploring Findings on Sexes and Age Groups
Abstract
Background: While medication errors (MEs) have been studied in the European Medicines Agency's EudraVigilance, extensive characterisation and signal detection based on sexes and age groups have not been attempted.
Objectives: The aim of this study was to characterise all ME-related individual case safety reports in EudraVigilance and explore notable signals of disproportionate reporting (SDRs) among sexes and age groups for the 30 most frequently reported drugs.
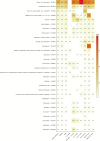
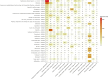
Methods: Individual case safety reports were used from EudraVigilance reported between 2002 and 2021. An ME was defined as any Preferred Term from the narrow Standardised Medical Dictionary for Regulatory Activities® Query. Signals of disproportionate reporting were selected based on a lower boundary of the 95% confidence interval ≥ 1 of the reporting odds ratio, and at least 3 individual case safety reports. Analysed subgroups were female individuals, male individuals, and age groups 0-1 month, 2 months to 2 years, 3-11 years, 12-17 years, 18-64 years, 65-85 years, and >85 years. Heatmaps were utilised as a visual aid to identify striking SDRs.
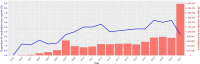
Results: Of the 9,662,345 EudraVigilance reports, 267,262 (2.8%) contained at least one ME, with a total of 300,324 MEs, for 429,554 drugs. The most reported ME was "Inappropriate schedule of product administration" (52,646; 17.5%), followed by "Incorrect dose administered" (32,379; 10.8%) and "Wrong technique in product usage process" (26,831; 8.9%). Individual case safety reports with MEs were most frequently related to female individuals (148,009; 55.4%), most often submitted by healthcare professionals (155,711; 58.3%), originated predominantly from the USA (98,716; 36.9%), followed by France (26,678; 10.0%), and showed a median reported age of 50 years (interquartile range: 26-68). Most ME individual case safety reports (158,991; 59.5%) were associated with a serious health outcome. A total of 847 SDRs were identified, based on the entire EudraVigilance database; for subgroups, the number of SDRs ranged from 84 for the age group 0-1 month to 749 for female individuals. Signals of disproportionate reporting for female individuals and male individuals were very similar. Most MEs were reported for the vaccine against human papillomavirus (Anatomical Therapeutic Chemical [ATC]: J07BM01; 11,086 MEs, 57% being "inappropriate schedule of product administration"), with reporting odds ratios that range from 1.5 to 47.0 among age groups. The SDR for the live-attenuated vaccine against herpes zoster (ATC: J07BK02) had a reporting odds ratio that ranged from 26.6 to 78.1 among all subgroups. Signals of disproportionate reporting for oxycodone (ATC: N02AA05; 847 cases of "Accidental overdose", 35%), risperidone (ATC: N05AX08; 469 cases "Inappropriate schedule of product administration", 22.3%) and rivaroxaban (ATC: B01AF01; 1,377 cases of "Incorrect dose administered", 34.6%) stood out with higher magnitude SDRs for the age group 2 months to 2 years, with an reporting odds ratio range between 8.2 and 10.7, while for the entire EudraVigilance the reporting odds ratio ranged between 1.3 and 1.6 for the same drugs.
Conclusions: This exploratory research provides an overview of characterised ME individual case safety reports and SDRs from the EudraVigilance database. Most conspicuous SDRs were identified in specific age groups. Signals of disproportionate reporting, not described in the literature, were found for vaccines, oxycodone, rivaroxaban and risperidone, and may prompt further examination by stakeholders. Top-reported MEs ("Inappropriate schedule of product administration", "Incorrect dose administered" and "Wrong technique in product usage process") emerged as a general priority focus to perform a further root-cause analysis involving healthcare providers, manufacturers and regulatory bodies, to improve the understanding and prevention of MEs.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Funding: This work has received support from the European Health Data & Evidence Network (EHDEN) project. EHDEN has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No. 806968. Conflicts of Interest/Competing Interests: The research group from the Department of Medical Informatics received unconditional research grants from Chiesi, UCB, Amgen, Johnson and Johnson, and the European Medicines Agency, none of which relates to the content of this article. Ethics Approval: Not applicable. Consent to Participate: Not applicable. Consent for Publication: Not applicable. Availability of Data and Material: An agreement has been made with the EMA not to transfer or dispose of the ICSR data set Level 2A for which access is provided under the condition of the Confidentiality Undertaking to any third party. It is agreed not to permit any third party to access, study, analyse, refer to or otherwise use the data or permit any party to reproduce any ICSR data. A publicly available version of this database is available here: https://www.adrreports.eu/en/index.html . Further inquiries can be directed to the corresponding author. Code Availability: Not applicable. Authors’ Contributions: VP: conceptualisation, formal analysis, visualisation, writing (original draft), writing (review and editing), methodology, visualisation. JK: software, writing (review and editing). EV: software, writing (review and editing). MdW: software, writing (review and editing). PR: supervision, writing (review and editing). KV: conceptualisation, supervision, writing (review and editing). All authors read and approved the final version.
Figures
Similar articles
-
Spontaneous reports of vaccination errors in the European regulatory database EudraVigilance: A descriptive study.Vaccine. 2018 Dec 18;36(52):7956-7964. doi: 10.1016/j.vaccine.2018.11.003. Epub 2018 Nov 8. Vaccine. 2018. PMID: 30416019
-
Descriptive analysis on disproportionate medication errors and associated patient characteristics in the Food and Drug Administration's Adverse Event Reporting System.Pharmacoepidemiol Drug Saf. 2024 Jan;33(1):e5743. doi: 10.1002/pds.5743. Epub 2023 Dec 29. Pharmacoepidemiol Drug Saf. 2024. PMID: 38158381
-
A characterization and disproportionality analysis of medication error related adverse events reported to the FAERS database.Expert Opin Drug Saf. 2018 Dec;17(12):1161-1169. doi: 10.1080/14740338.2018.1550069. Epub 2018 Nov 27. Expert Opin Drug Saf. 2018. PMID: 30451017
-
Evaluation of quantitative signal detection in EudraVigilance for orphan drugs: possible risk of false negatives.Ther Adv Drug Saf. 2019 Oct 21;10:2042098619882819. doi: 10.1177/2042098619882819. eCollection 2019. Ther Adv Drug Saf. 2019. PMID: 31673326 Free PMC article. Review.
-
Evaluation of patient reporting of adverse drug reactions to the UK 'Yellow Card Scheme': literature review, descriptive and qualitative analyses, and questionnaire surveys.Health Technol Assess. 2011 May;15(20):1-234, iii-iv. doi: 10.3310/hta15200. Health Technol Assess. 2011. PMID: 21545758 Review.
Cited by
-
Exploring the Correlation Between Patient Safety Culture and Adverse Medical Events Using Failure Mode and Effect Analysis (FMEA).Risk Manag Healthc Policy. 2025 Apr 18;18:1367-1376. doi: 10.2147/RMHP.S502725. eCollection 2025. Risk Manag Healthc Policy. 2025. PMID: 40264504 Free PMC article.
References
-
- EMA. Medication errors 2021. Available from: https://www.ema.europa.eu/en/human-regulatory/post-authorisation/pharmac.... Accessed 2 Sep 2024.
-
- Pera V, van Vaerenbergh F, Kors JA, van Mulligen EM, Parry R, de Wilde M, et al. Descriptive analysis on disproportionate medication errors and associated patient characteristics in the Food and Drug Administration’s Adverse Event Reporting System. Pharmacoepidemiol Drug Saf. 10.1002/pds.5743. - PubMed
-
- Mirosevic Skvrce N, Galic I, Pacadi C, Kandzija N, Mucalo I. Adverse drug reactions that arise from the use of medicinal products outside the terms of the marketing authorisation. Res Soc Admin Pharm. 2020;16:928–34. 10.1016/j.sapharm.2019.10.003. - PubMed
-
- Assiri GA, Shebl NA, Mahmoud MA, Aloudah N, Grant E, Aljadhey H, et al. What is the epidemiology of medication errors, error-related adverse events and risk factors for errors in adults managed in community care contexts? A systematic review of the international literature. BMJ Open. 2018;8: e019101. 10.1136/bmjopen-2017-019101. - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

