Safety and efficacy of apixaban versus warfarin in peritoneal dialysis patients with non-valvular atrial fibrillation: protocol for a prospective, randomised, open-label, blinded endpoint trial (APIDP2)
- PMID: 39306346
- PMCID: PMC11535701
- DOI: 10.1136/bmjopen-2024-089353
Safety and efficacy of apixaban versus warfarin in peritoneal dialysis patients with non-valvular atrial fibrillation: protocol for a prospective, randomised, open-label, blinded endpoint trial (APIDP2)
Abstract
Introduction: Several randomised controlled trials have demonstrated that novel oral anticoagulants are safer compared with vitamin K antagonists for the management of non-valvular atrial fibrillation (NVAF) to prevent thromboembolic events in the general population. There is a growing interest in the use of apixaban in patients with end-stage renal disease (ESRD) undergoing peritoneal dialysis (PD) but there is a lack of randomised data in this population.
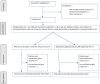
Methods and analysis: APIDP2 is a prospective parallel, randomised, open-label, blinded endpoint trial involving patients with ESRD undergoing chronic PD who have NVAF. A total of 178 participants will be recruited from 20 French PD centres. Eligible patients will be randomly assigned to receive either apixaban at a reduced dose of 2.5 mg two times per day (dose determined with the previous pharmacokinetic study APIDP1) or dose-adjusted to international normalised ratio (INR) target (2-3) coumadin therapy. Anticoagulation to prevent thromboembolic events will be initiated or changed according to the randomisation for a duration of 1 year. The primary outcome is a major or clinically relevant non-major bleeding from randomisation up to month 12, assessed according to the International Society on Thrombosis and Haemostasis Score. Secondary outcomes encompass an efficacy composite criterion combining stroke or transient ischaemic attack (TIA), cardiovascular death and thrombosis including myocardial infarction cumulated at 12 months. Bleeding events will be also classified according to Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO) and Thrombolysis In Myocardial Infarction (TIMI) criteria and pharmacodynamics outcomes will evaluate the time within the INR target range of 2-3 in the warfarin arm over 1 year, and anti-Xa apixaban activity in case of bleeding events and at 1 month, 6 months and 12 months of follow-up in the apixaban arm. To demonstrate that apixaban is safer than warfarin at 1 year, assuming two interim analyses after 60 and 118 patients, a bilateral alpha risk of 5% and a power of 80%, 178 patients are needed in this randomised trial (effect size found from the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation (ARISTOTLE) Study among patients with creatinine clearance 25-30 ml/min), that is, 89 patients per group.
Ethics and dissemination: The study has been approved by the ethics committee Comité de Protection des Personnes Sud Est III - Lyon - FRANCE, CT number 2023-507544-37-00. Written informed consent is required for each participant. Findings will be presented at scientific meetings and published in peer-reviewed journals.
Trial registration: ClinicalTrials.gov, NCT06045858; European Clinical Trial System, CT number 2023-507544-37-00.
Keywords: Anticoagulation; Dialysis; Randomized Controlled Trial.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials