Fluorescence-plane polarization for the real-time monitoring of transferase migration in living cells
- PMID: 39309085
- PMCID: PMC11409853
- DOI: 10.1039/d4sc03387f
Fluorescence-plane polarization for the real-time monitoring of transferase migration in living cells
Abstract
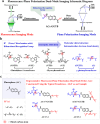
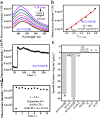
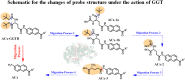
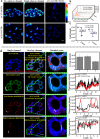
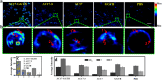
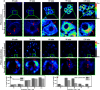
Transferases are enzymes that exhibit multisite migration characteristics. Significantly, enzyme activity undergoes changes during this migration process, which inevitably impacts the physiological function of living organisms and can even lead to related malignant diseases. However, research in this field has been severely hindered by the lack of tools for the simultaneous and differential monitoring of site-specific transferase activity. Herein, we propose a novel strategy that integrates a fluorescence signal response with high sensitivity and an optical rotation signal response with superior spatial resolution. To validate the feasibility of this strategy, transferase γ-glutamyltransferase (GGT) was used as a model system to develop dual-mode chiral probes ACx-GGTB (AC17-GGTB and AC15-GGTB) using chiral amino acids as specific bifunctional recognition groups. The probes undergo structural changes under GGT, resulting in the release of bifunctional recognition groups (chiral amino acids) and simultaneously generate fluorescence signals and optical rotation signals. This dual-mode output exhibits high sensitivity and facilitates differentiation of sites. Furthermore, it enables simultaneous and differential detection of GGT activity at different sites during migration. We anticipate that probes developed based on this strategy will facilitate imaging-based monitoring of the activity for other transferases, thus providing an imaging platform suitable for the real-time tracking of transferase activity changes during migration.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures


References
LinkOut - more resources
Full Text Sources
Miscellaneous

