Nanoscale ZnO doping in prosthetic polymers mitigate wear particle-induced inflammation and osteolysis through inhibiting macrophage secretory autophagy
- PMID: 39309162
- PMCID: PMC11415586
- DOI: 10.1016/j.mtbio.2024.101225
Nanoscale ZnO doping in prosthetic polymers mitigate wear particle-induced inflammation and osteolysis through inhibiting macrophage secretory autophagy
Abstract
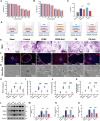
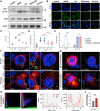
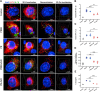
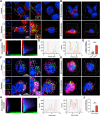
Wear particles produced by joint replacements induce inflammatory responses that lead to periprosthetic osteolysis and aseptic loosening. However, the precise mechanisms driving wear particle-induced osteolysis are not fully understood. Recent evidence suggests that autophagy, a cellular degradation process, plays a significant role in this pathology. This study aimed to clarify the role of autophagy in mediating inflammation and osteolysis triggered by wear particles and to evaluate the therapeutic potential of zinc oxide nanoparticles (ZnO NPs). We incorporated ZnO into the prosthetic material itself, ensuring that the wear particles inherently carried ZnO, providing a targeted and sustained intervention. Our findings reveal that polymer wear particles induce excessive autophagic activity, which is closely associated with increased inflammation and osteolysis. We identified secretory autophagy as a key mechanism for IL-1β secretion, exacerbating osteolysis. Both in vitro and in vivo experiments demonstrated that ZnO-doped particles significantly inhibit autophagic overactivation, thereby reducing inflammation and osteolysis. In summary, this study establishes secretory autophagy as a critical mechanism in wear particle-induced osteolysis and highlights the potential of ZnO-doped prosthetic polymers for targeted, sustained mitigation of periprosthetic osteolysis.
Keywords: Aseptic loosening; Macrophages; Osteoclastogenesis; Periprosthetic osteolysis; Secretory autophagy; Wear particle-induced inflammation.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





References
LinkOut - more resources
Full Text Sources

