Age-induced changes in skeletal muscle mitochondrial DNA synthesis, quantity, and quality in genetically unique rats
- PMID: 39312152
- PMCID: PMC11872842
- DOI: 10.1007/s11357-024-01344-4
Age-induced changes in skeletal muscle mitochondrial DNA synthesis, quantity, and quality in genetically unique rats
Abstract
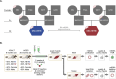
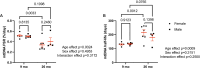
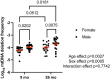
Mitochondrial genomic integrity is a key element of physiological processes and health. Changes in the half-life of the mitochondrial genome are implicated in the generation and accumulation of age-induced mitochondrial DNA (mtDNA) mutations, which are implicated in skeletal muscle aging and sarcopenia. There are conflicting data on the half-life of mtDNA, and there is limited information on how aging affects half-life in skeletal muscle. We hypothesized that skeletal muscle mtDNA synthesis rates would decrease with age in both female and male rats concomitant with changes in mtDNA integrity reflected in mtDNA copy number and mutation frequency. We measured mitochondrial genome half-life using stable isotope labeling over a period of 14 days and assessed mtDNA copy number and deletion mutation frequency using digital PCR in the quadriceps muscle of 9-month-old and 26-month-old male and female OKC-HET rats. We found a significant age-related increase in mtDNA half-life, from 132 days at 9 months to 216 days at 26 months of age in OKC-HET quadriceps. Concomitant with the increase in mtDNA half-life, we found an age-related increase in mtDNA deletion mutation frequency in both male and female rats. Notably, 26-month-old female rats had a lower mutation frequency than male rats, and there were no changes in mtDNA copy number with sex, age, or mitochondrial genotype. These data reveal several key findings: (1) mtDNA turnover in rat skeletal muscle decreases with age, (2) mtDNA half-lives in skeletal muscle are approximately an order of magnitude longer than what is reported for other tissues, and (3) muscle mtDNA turnover differs significantly from the turnover of other mitochondrial macromolecules including components of the mitochondrial nucleoid. These findings provide insight into the factors driving age-induced mtDNA mutation accumulation, which contribute to losses of mitochondrial genomic integrity and may play a role in skeletal muscle dysfunction.
Keywords: Aging; Deuterium oxide; Mitochondrial DNA; Mutation; Rats; Skeletal muscle.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures


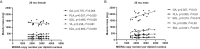
Similar articles
-
Skeletal muscle mitochondrial DNA copy number and mitochondrial DNA deletion mutation frequency as predictors of physical performance in older men and women.Geroscience. 2021 Jun;43(3):1253-1264. doi: 10.1007/s11357-021-00351-z. Epub 2021 Mar 19. Geroscience. 2021. PMID: 33740224 Free PMC article.
-
Mitochondrial DNA deletion mutations increase exponentially with age in human skeletal muscle.Aging Clin Exp Res. 2021 Jul;33(7):1811-1820. doi: 10.1007/s40520-020-01698-7. Epub 2020 Sep 23. Aging Clin Exp Res. 2021. PMID: 32965609 Free PMC article.
-
Loss of the antioxidant enzyme CuZnSOD (Sod1) mimics an age-related increase in absolute mitochondrial DNA copy number in the skeletal muscle.Age (Dordr). 2016 Aug;38(4):323-333. doi: 10.1007/s11357-016-9930-1. Epub 2016 Jul 21. Age (Dordr). 2016. PMID: 27444179 Free PMC article.
-
Tissue mitochondrial DNA changes. A stochastic system.Ann N Y Acad Sci. 2000 Jun;908:226-43. doi: 10.1111/j.1749-6632.2000.tb06650.x. Ann N Y Acad Sci. 2000. PMID: 10911962 Review.
-
Mitochondria, muscle health, and exercise with advancing age.Physiology (Bethesda). 2015 May;30(3):208-23. doi: 10.1152/physiol.00039.2014. Physiology (Bethesda). 2015. PMID: 25933821 Review.
Cited by
-
Mitochondrial dysfunction in the regulation of aging and aging-related diseases.Cell Commun Signal. 2025 Jun 19;23(1):290. doi: 10.1186/s12964-025-02308-7. Cell Commun Signal. 2025. PMID: 40537801 Free PMC article. Review.
References
-
- Lopez-Otin C, et al. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243–78. - PubMed
-
- Kovalchuk O, Kovalchuk I. Genome stability: from virus to human application. Translational epigenetics series. Amsterdam: Elsevier/Academic Press; 2016. xxii, pp. 690.
-
- Vijg J, Suh Y. Genome instability and aging. Annu Rev Physiol. 2013;75:645–68. - PubMed
-
- Panier S, Wang S, Schumacher B. Genome instability and DNA repair in somatic and reproductive aging. Annu Rev Pathol. 2024;19:261–90. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

