Activation Pathways of Murine Macrophages by Lipophosphoglycan from Strains of Leishmania major (FV1 and LV39)
- PMID: 39313410
- PMCID: PMC11474971
- DOI: 10.1021/acsinfecdis.4c00295
Activation Pathways of Murine Macrophages by Lipophosphoglycan from Strains of Leishmania major (FV1 and LV39)
Abstract
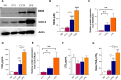
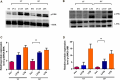
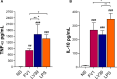
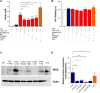
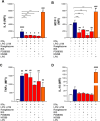
Lipophosphoglycan (LPG) is an important Leishmania virulence factor. It is the most abundant surface glycoconjugate in promastigotes, playing an important role in the interaction with phagocytic cells. While LPG is known to modulate the macrophage immune response during infection, the activation mechanisms triggered by this glycoconjugate have not been fully elucidated. This work investigated the role that LPGs purified from two strains of Leishmania major (FV1 and LV39) play in macrophage activation, considering the differences in their biochemical structures. Bone marrow-derived macrophages from BALB/c mice were stimulated with 10 μg/mL purified LPG from the LV39 and FV1 strains. We then measured the production of nitric oxide (NO) and cytokines, the expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), and the activation of MAPK pathways. LPG from the LV39 strain, which has longer poly-galactosylated side chains, induced a more pro-inflammatory profile than that from the FV1 strain. This included higher production of NO, TNF-α, and PGE2, and increased expression of COX-2 and iNOS. Additionally, the phosphorylation of ERK-1/2 and JNK was elevated in macrophages exposed to LPG from the LV39 strain. No difference in IL-10 production was observed in cells stimulated by both LPG. Thus, intraspecific structural differences in LPG contribute to distinct innate immune responses in macrophages.
Keywords: Leishmania major; Lipophosphoglycan; cytokines; inflammatory mediators; leishmaniasis; macrophage.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Two biochemically distinct lipophosphoglycans from Leishmania braziliensis and Leishmania infantum trigger different innate immune responses in murine macrophages.Parasit Vectors. 2013 Mar 7;6:54. doi: 10.1186/1756-3305-6-54. Parasit Vectors. 2013. PMID: 23497381 Free PMC article.
-
Intraspecies Polymorphisms in the Lipophosphoglycan of L. braziliensis Differentially Modulate Macrophage Activation via TLR4.Front Cell Infect Microbiol. 2019 Jul 10;9:240. doi: 10.3389/fcimb.2019.00240. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31355149 Free PMC article.
-
Leishmania expressed lipophosphoglycan interacts with Toll-like receptor (TLR)-2 to decrease TLR-9 expression and reduce anti-leishmanial responses.Clin Exp Immunol. 2013 Jun;172(3):403-9. doi: 10.1111/cei.12074. Clin Exp Immunol. 2013. PMID: 23600828 Free PMC article.
-
Adversarial relationship between the leishmania lipophosphoglycan and protein kinase C of host macrophages.Parasite Immunol. 1999 Dec;21(12):597-600. doi: 10.1046/j.1365-3024.1999.00266.x. Parasite Immunol. 1999. PMID: 10583861 Review.
-
Cytokines and nitric oxide as effector molecules against parasitic infections.Philos Trans R Soc Lond B Biol Sci. 1997 Sep 29;352(1359):1311-5. doi: 10.1098/rstb.1997.0115. Philos Trans R Soc Lond B Biol Sci. 1997. PMID: 9355122 Free PMC article. Review.
References
-
- Favila M. A.; Geraci N. S.; Jayakumar A.; Hickerson S.; Mostrom J.; Turco S. J.; Beverley S. M.; McDowell M. A. Differential Impact of LPG-and PG-Deficient Leishmania Major Mutants on the Immune Response of Human Dendritic Cells. PLoS Neglected Trop. Dis. 2015, 9 (12), e000423810.1371/journal.pntd.0004238. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

