The immunoglobulin M-degrading enzyme of Streptococcus suis (IdeSsuis) leads to long-lasting inhibition of the activation of porcine IgM-secreting B cells
- PMID: 39313819
- PMCID: PMC11421183
- DOI: 10.1186/s13567-024-01363-1
The immunoglobulin M-degrading enzyme of Streptococcus suis (IdeSsuis) leads to long-lasting inhibition of the activation of porcine IgM-secreting B cells
Abstract
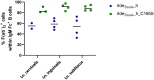
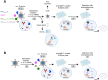
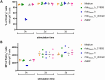
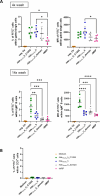
Streptococcus suis (S. suis) is one of the most important porcine pathogens, causing severe pathologies such as meningitis or polyarthritis. It is also a very successful colonizer of mucosal surfaces. The IgM-degrading enzyme of S. suis (IdeSsuis) specifically cleaves porcine IgM, which results in complement evasion. On the basis of our previous finding that IdeSsuis also cleaves the IgM B cell receptor in vitro, we verified IgM B cell receptor cleavage ex vivo in whole regional lymph nodes and investigated the working hypothesis that this IgM B cell receptor cleavage results in a long-lasting impaired B cell function. The number of IgM-secreting cells was determined via ELISpot analysis after porcine peripheral blood mononuclear cells had initially been treated with different recombinant S. suis proteins and subsequently stimulated with interleukin-2 and the toll-like receptor 7/8 ligand R848. Compared with treatment with medium or recombinant muramidase-released protein, treatment with rIdeSsuis but also with a cleavage-deficient variant led to a reduction in the number of IgM-secreting cells as well as the level of secreted IgM. Flow cytometry analysis confirmed that the IgM B cell receptor was cleaved only by rIdeSsuis, and the receptor recovered to pretreatment levels on day 2 after treatment. Flow cytometry analysis of B and T cells incubated with fluorescein-labelled recombinant proteins revealed that different rIdeSsuis variants bind specifically to B cells, most prominently the cleavage-deficient variant. Our results indicate that in vitro interference of rIdeSsuis with the IgM B cell receptor results in long-lasting impaired IgM secretion by B cells after toll-like receptor activation. Further studies are warranted to prove that the modulation of B cell function by IdeSsuis could play a role in vivo.
Keywords: Streptococcus suis; B cell receptor; B cell receptor cleavage; ELISpot; IdeSsuis; IgM.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
IgM cleavage by Streptococcus suis reduces IgM bound to the bacterial surface and is a novel complement evasion mechanism.Virulence. 2018;9(1):1314-1337. doi: 10.1080/21505594.2018.1496778. Virulence. 2018. PMID: 30001174 Free PMC article.
-
The immunoglobulin M-degrading enzyme of Streptococcus suis, IdeSsuis, is a highly protective antigen against serotype 2.Vaccine. 2015 May 5;33(19):2207-2212. doi: 10.1016/j.vaccine.2015.03.047. Epub 2015 Mar 28. Vaccine. 2015. PMID: 25825330
-
Immunoglobulin M-degrading enzyme of Streptococcus suis (Ide Ssuis ) impairs porcine B cell signaling.Front Immunol. 2023 Feb 16;14:1122808. doi: 10.3389/fimmu.2023.1122808. eCollection 2023. Front Immunol. 2023. PMID: 36875121 Free PMC article.
-
Meningitis in pigs caused by Streptococcus suis--a speculative review.Vet Microbiol. 1993 Jul;36(1-2):39-55. doi: 10.1016/0378-1135(93)90127-s. Vet Microbiol. 1993. PMID: 8236779 Review. No abstract available.
-
Review of the speculative role of co-infections in Streptococcus suis-associated diseases in pigs.Vet Res. 2021 Mar 20;52(1):49. doi: 10.1186/s13567-021-00918-w. Vet Res. 2021. PMID: 33743838 Free PMC article. Review.
References
-
- Gottschalk M, Segura M (2019) Streptococcosis. In: Zimmerman JJ, Karriker LA, Ramirez A, Schwartz KJ, Stevenson GW, Zhang J (eds) Disease of swine, 11th edn. Wiley-Blackwell, Hoboken NJ, pp 934–950
-
- Baele M, Chiers K, Devriese LA, Smith HE, Wisselink HJ, Vaneechoutte M, Haesebrouck F (2001) The gram-positive tonsillar and nasal flora of piglets before and after weaning. J Appl Microbiol 91:997–1003 - PubMed
-
- Wertheim HFL, Nghia HDT, Taylor W, Schultsz C (2009) Streptococcus suis: an emerging human pathogen. Clin Infect Dis 48:617–625 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

