This is a preprint.
Epidermal Growth Factor Receptor Signaling Governs the Host Inflammatory Response to Invasive Aspergillosis
- PMID: 39314401
- PMCID: PMC11419056
- DOI: 10.1101/2024.09.10.612305
Epidermal Growth Factor Receptor Signaling Governs the Host Inflammatory Response to Invasive Aspergillosis
Update in
-
Epidermal growth factor receptor signaling governs the host inflammatory response to invasive aspergillosis.mBio. 2024 Dec 11;15(12):e0267124. doi: 10.1128/mbio.02671-24. Epub 2024 Oct 30. mBio. 2024. PMID: 39475281 Free PMC article.
Abstract
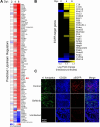
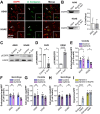
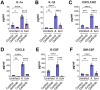
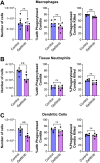
The epidermal growth factor receptor (EGFR) has been identified as an epithelial cell receptor for Mucorales fungi and Candida albicans. Blocking EGFR with small molecule inhibitors reduces disease severity in mouse models of mucormycosis and oropharyngeal candidiasis. In contrast, cases of invasive aspergillosis have been reported in cancer patients who were treated with EGFR inhibitors, suggesting that EGFR signaling may play a protective role in the host defense against this infection. Here, we analyzed transcriptomic data from the lungs of mice with invasive aspergillosis and found evidence that Aspergillus fumigatus infection activates multiple genes that are predicted to function in the EGFR signaling pathway. We also found that A. fumigatus infection activates EGFR in both a human small airway epithelial (HSAE) cell line and in the lungs of immunosuppressed mice. EGFR signaling in HSAE cells is required for maximal endocytosis of A. fumigatus and for fungal-induced proinflammatory cytokine and chemokine production. In a corticosteroid immunosuppressed mouse model of invasive pulmonary aspergillosis, inhibition of EGFR with gefitinib decreased whole lung chemokine levels and reduced accumulation of phagocytes in the lung, leading to a decrease in fungal killing, an increase in pulmonary fungal burden, and accelerated mortality. Thus, EGFR signaling is required for pulmonary epithelial cells to orchestrate the host innate immune defense against invasive aspergillosis in immunosuppressed hosts.
Keywords: Aspergillus fumigatus; alveolar epithelial cell; chemokine; cytokine; endocytosis; epidermal growth factor receptor; small airway epithelial cell.
Figures

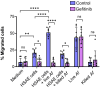
References
-
- Maertens JA, Rahav G, Lee DG, Ponce-de-Leon A, Ramirez Sanchez IC, Klimko N, Sonet A, Haider S, Diego Velez J, Raad I, Koh LP, Karthaus M, Zhou J, Ben-Ami R, Motyl MR, Han S, Grandhi A, Waskin H, study i. 2021. Posaconazole versus voriconazole for primary treatment of invasive aspergillosis: a phase 3, randomised, controlled, non-inferiority trial. Lancet 397:499–509. - PubMed
-
- Maertens JA, Raad II, Marr KA, Patterson TF, Kontoyiannis DP, Cornely OA, Bow EJ, Rahav G, Neofytos D, Aoun M, Baddley JW, Giladi M, Heinz WJ, Herbrecht R, Hope W, Karthaus M, Lee DG, Lortholary O, Morrison VA, Oren I, Selleslag D, Shoham S, Thompson GR 3rd, Lee M, Maher RM, Schmitt-Hoffmann AH, Zeiher B, Ullmann AJ. 2016. Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): a phase 3, randomised-controlled, non-inferiority trial. Lancet 387:760–9. - PubMed
-
- Resendiz-Sharpe A, Mercier T, Lestrade PPA, van der Beek MT, von dem Borne PA, Cornelissen JJ, De Kort E, Rijnders BJA, Schauwvlieghe A, Verweij PE, Maertens J, Lagrou K. 2019. Prevalence of voriconazole-resistant invasive aspergillosis and its impact on mortality in haematology patients. J Antimicrob Chemother 74:2759–2766. - PubMed
-
- Bertuzzi M, Schrettl M, Alcazar-Fuoli L, Cairns TC, Munoz A, Walker LA, Herbst S, Safari M, Cheverton AM, Chen D, Liu H, Saijo S, Fedorova ND, Armstrong-James D, Munro CA, Read ND, Filler SG, Espeso EA, Nierman WC, Haas H, Bignell EM. 2014. The pH-responsive PacC transcription factor of Aspergillus fumigatus governs epithelial entry and tissue invasion during pulmonary aspergillosis. PLoS Pathog 10:e1004413. - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
