A new mechanism of cancer initiation that involves the transformation of hepatocytes into preneoplastic single hepatocytes and minifoci positive for glutathione S-transferase P-form (GST-P) in rat livers: 3D analysis using a vibratome
- PMID: 39318029
- PMCID: PMC11422180
- DOI: 10.1002/cam4.70165
A new mechanism of cancer initiation that involves the transformation of hepatocytes into preneoplastic single hepatocytes and minifoci positive for glutathione S-transferase P-form (GST-P) in rat livers: 3D analysis using a vibratome
Abstract
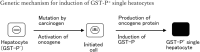
Background: Cancer initiation has long been "unknowable" in biology and medicine. In 1987, however, Moore and our research group observed single hepatocytes and minifoci that were strongly positive for glutathione S-transferase P-form (GST-P) in the rat liver as early as 2 to 3 days after initiation by diethylnitrosamine prior to the induction of GST-P+ foci and nodules. The induction of GST-P+ single hepatocytes, precursors of GST-P+ foci and nodules, was considered genetic. But, the details of the induction mechanism have remained unclear despite various examinations over a long period.
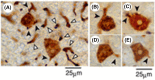
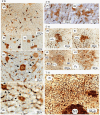
Methods: Male Sprague-Dawley rats (aged 6 weeks) were fed a basal diet containing either benzyl isothiocyanate (BITC, 0.5% by wt) or 2-acetylaminofluorene (AAF, 0.04%) ad libitum for appropriate time intervals. All animals were anesthetized and euthanized. The livers obtained were excised, cut into 3- to 4-mm-thick slices and fixed in cold acetone at 4 °C. The liver specimens were then sliced into 25-µm-thick sections in PBS using an automated microtome (Vibratome 1500 Sectioning System, Vibratome Products, NY, USA). Immunocytochemical staining was performed in free solution, and the results were examined via digital light microscopy (Coolscope, Nikon, Tokyo).
Results: 3D analysis using a vibratome showed that GST-P is rapidly excreted into the bile of the liver of animals in response to strong carcinogenic stress caused by promoters or initiators. "Rapid biliary excretion of GST-P" was widely and commonly observed in all hepatocytes, GST-P+ single hepatocytes, minifoci, foci and nodules under appropriate conditions. Surprisingly, on the basis of these key findings, a new mechanism of cancer initiation involving the transformation of hepatocytes into GST-P+ single hepatocytes and minifoci in animal livers was identified. In addition, the initiation process was determined to be nongenetic because mutation is an invisible rare event.
Conclusions: This short review describes several details about breakthrough findings on cancer initiation in rat livers, the application of 3D analysis to other cancers and the importance in the genetic analysis in malignant diseases.
Keywords: 3D analysis; GST‐P; cancer initiation; chemical hepatocarcinogenesis; tumor markers; vibratome.
© 2024 The Author(s). Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
The author declares no conflicts of interest to disclose.
Figures



Similar articles
-
Strong carcinogenic stress response induction of preneoplastic cells positive for GST-P in the rat liver: Physiological mechanism for initiation.Life Sci. 2018 May 1;200:42-48. doi: 10.1016/j.lfs.2018.02.041. Epub 2018 Mar 2. Life Sci. 2018. PMID: 29501922
-
Vibratome technique revealed initial carcinogenic changes that induce GST-P+ single hepatocytes and minifoci in rat liver.Anal Biochem. 2023 Jul 1;672:115168. doi: 10.1016/j.ab.2023.115168. Epub 2023 Apr 18. Anal Biochem. 2023. PMID: 37080414
-
Bile duct-bound growth of precursor cells of preneoplastic foci inducible in the initiation stage of rat chemical hepatocarcinogenesis by 2-acetylaminofluorene.Jpn J Clin Oncol. 2008 Sep;38(9):604-10. doi: 10.1093/jjco/hyn080. Epub 2008 Aug 16. Jpn J Clin Oncol. 2008. PMID: 18708648
-
Value of GST-P positive preneoplastic hepatic foci in dose-response studies of hepatocarcinogenesis: evidence for practical thresholds with both genotoxic and nongenotoxic carcinogens. A review of recent work.Toxicol Pathol. 2003 Jan-Feb;31(1):80-6. doi: 10.1080/01926230390173879. Toxicol Pathol. 2003. PMID: 12597451 Review.
-
Is placental glutathione S-Transferase (GST-P) a suitable biomarker for oral carcinogenesis: A scoping review.Pathol Res Pract. 2023 Sep;249:154762. doi: 10.1016/j.prp.2023.154762. Epub 2023 Aug 16. Pathol Res Pract. 2023. PMID: 37657165
References
-
- Boveri T. The Origin of Malignant Tumors. Williams & Wilkins; 1929:62‐63.
-
- Pitot HC. Principles of carcinogenesis. Chemical. In: VT DV Jr, Hellman SA, Rosenburg S, eds. Cancer: Principles and Practice of Oncology. 2nd ed. Lippincott JB; 1985:116‐135.
-
- Prehn RT. Cancers beget mutations versus mutations beget cancers. Cancer Res. 1994;54:5296‐5300. - PubMed
-
- Soto AM, Sonnenschein C. The somatic mutation theory of cancer: growing problems with the paradigm? BioEssays. 2004;26:1097‐1107. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- Hirosaki University Fund for Promotion of International Scientific Research (Hirosaki, Japan)
- 07457579/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan
- 60570104/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan
LinkOut - more resources
Full Text Sources
Research Materials

