Towards tunable exciton delocalization in DNA Holliday junction-templated indodicarbocyanine 5 (Cy5) dye derivative heterodimers
- PMID: 39320147
- PMCID: PMC11423794
- DOI: 10.1039/d4nh00225c
Towards tunable exciton delocalization in DNA Holliday junction-templated indodicarbocyanine 5 (Cy5) dye derivative heterodimers
Abstract
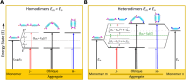
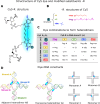
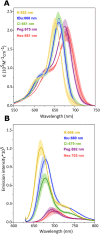
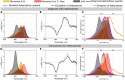
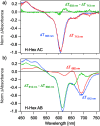
We studied the exciton delocalization of indodicarbocyanine 5 dye derivative (Cy5-R) heterodimers templated by a DNA Holliday junction (HJ), which was quantified by the exciton hopping parameter Jm,n. These dyes were modified at the 5 and 5' positions of indole rings with substituent (R) H, Cl, tBu, Peg, and hexyloxy (Hex) groups that exhibit different bulkiness and electron-withdrawing/donating capacities. The substituents tune the physical properties of the dyes, such as hydrophobicity (log P) and solvent-accessible surface area (SASA). We tuned the Jm,n of heterodimers by attaching two Cy5-Rs in adjacent and transverse positions along the DNA-HJ. Adjacent heterodimers exhibited smaller Jm,n compared to transverse heterodimers, and some adjacent heterodimers displayed a mixture of H- and J-like aggregates. Most heterodimers exhibited Jm,n values within the ranges of the corresponding homodimers, but some heterodimers displayed synergistic exciton delocalization that resulted in larger Jm,n compared to their homodimers. We then investigated how chemically distinct Cy5-R conjugated to DNA can interact to create delocalized excitons. We determined that heterodimers involving Cy5-H and Cy5-Cl and a dye with larger substituents (bulky substituents and large SASA) such as Cy5-Peg, Cy5-Hex, and Cy5-tBu resulted in larger Jm,n. The combination provides steric hindrance that optimizes co-facial packing (bulky Cy5-R) with a smaller footprint (small SASA) that maximizes proximity. The results of this study lay a groundwork for rationally optimizing the exciton delocalization in dye aggregates for developing next-generation technologies based on optimized exciton transfer efficiency such as quantum information systems and biomedicine.
Conflict of interest statement
There are no conflicts to declare.
Figures


Similar articles
-
Effect of hydrophilicity-imparting substituents on exciton delocalization in squaraine dye aggregates covalently templated to DNA Holliday junctions.Nanoscale. 2024 Jan 18;16(3):1206-1222. doi: 10.1039/d3nr04499h. Nanoscale. 2024. PMID: 38113123
-
Towards control of excitonic coupling in DNA-templated Cy5 aggregates: the principal role of chemical substituent hydrophobicity and steric interactions.Nanoscale. 2023 Feb 16;15(7):3284-3299. doi: 10.1039/d2nr05544a. Nanoscale. 2023. PMID: 36723027 Free PMC article.
-
Exciton Delocalization in Indolenine Squaraine Aggregates Templated by DNA Holliday Junction Scaffolds.J Phys Chem B. 2020 Oct 29;124(43):9636-9647. doi: 10.1021/acs.jpcb.0c06480. Epub 2020 Oct 14. J Phys Chem B. 2020. PMID: 33052691 Free PMC article.
-
Large Davydov Splitting and Strong Fluorescence Suppression: An Investigation of Exciton Delocalization in DNA-Templated Holliday Junction Dye Aggregates.J Phys Chem A. 2018 Mar 1;122(8):2086-2095. doi: 10.1021/acs.jpca.7b12668. Epub 2018 Feb 19. J Phys Chem A. 2018. PMID: 29420037 Free PMC article.
-
Fluorescence resonance energy transfer (FRET) and competing processes in donor-acceptor substituted DNA strands: a comparative study of ensemble and single-molecule data.J Biotechnol. 2002 Jan;82(3):211-31. doi: 10.1016/s1389-0352(01)00039-3. J Biotechnol. 2002. PMID: 11999691 Review.
Cited by
-
Distinguishing packing configurations of molecular dimers using excited-state absorption peaks in two-dimensional electronic spectra.J Chem Phys. 2025 May 7;162(17):174303. doi: 10.1063/5.0258934. J Chem Phys. 2025. PMID: 40309947
References
-
- Yurke B. Kuang W. Phys. Rev. A: At., Mol., Opt. Phys. 2010;81:033814. doi: 10.1103/PhysRevA.81.033814. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

