Balancing functions of antifouling, nitric oxide release and vascular cell selectivity for enhanced endothelialization of assembled multilayers
- PMID: 39323744
- PMCID: PMC11422184
- DOI: 10.1093/rb/rbae096
Balancing functions of antifouling, nitric oxide release and vascular cell selectivity for enhanced endothelialization of assembled multilayers
Abstract
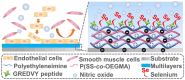
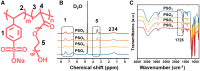
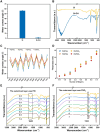
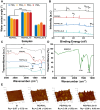
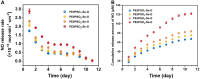
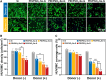
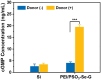
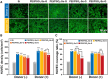
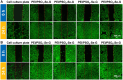
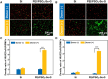
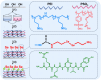
Surface endothelialization is a promising way to improve the hemocompatibility of biomaterials. However, current surface endothelialization strategies have limitations. For example, various surface functions are not well balanced, leading to undesirable results, especially when multiple functional components are introduced. In this work, a multifunctional surface was constructed by balancing the functions of antifouling, nitric oxide (NO) release and endothelial cell promotion via layer-by-layer (LBL) self-assembly. Poly(sodium p-styrenesulfonate-co-oligo(ethylene glycol) methacrylate) (negatively charged) and polyethyleneimine (positively charged) were deposited on silicon substrates to construct multilayers by LBL self-assembly. Then, organic selenium, which has a NO-releasing function, and the cell-adhesive peptide Gly-Arg-Glu-Asp-Val-Tyr, which selectively promotes endothelial cells, were introduced on the assembled multilayers. Poly(oligo(ethylene glycol) methacrylate) is a hydrophilic component for antifouling properties, and poly(sodium p-styrenesulfonate) is a heparin analog that provides negative charges. By modulating the contents of poly(oligo(ethylene glycol) methacrylate) and poly(sodium p-styrenesulfonate) in the copolymers, the NO release rates catalyzed by the modified surfaces were regulated. Moreover, the behaviors of endothelial cells and smooth muscle cells on modified surfaces were well controlled. The optimized surface strongly promoted endothelial cells and inhibited smooth muscle cells to achieve endothelialization effectively.
Keywords: endothelialization; heparin analogs; layer-by-layer self-assembly; nitric oxide release; surface modification.
© The Author(s) 2024. Published by Oxford University Press.
Figures




Similar articles
-
Multistage Anticoagulant Surfaces: A Synergistic Combination of Protein Resistance, Fibrinolysis, and Endothelialization.ACS Appl Mater Interfaces. 2023 Aug 2;15(30):35860-35871. doi: 10.1021/acsami.3c05145. Epub 2023 Jul 19. ACS Appl Mater Interfaces. 2023. PMID: 37466472
-
Co-immobilization of ACH11 antithrombotic peptide and CAG cell-adhesive peptide onto vascular grafts for improved hemocompatibility and endothelialization.Acta Biomater. 2019 Oct 1;97:344-359. doi: 10.1016/j.actbio.2019.07.057. Epub 2019 Aug 1. Acta Biomater. 2019. PMID: 31377424
-
Nanofibrous heparin and heparin-mimicking multilayers as highly effective endothelialization and antithrombogenic coatings.Biomacromolecules. 2015 Mar 9;16(3):992-1001. doi: 10.1021/bm501882b. Epub 2015 Feb 20. Biomacromolecules. 2015. PMID: 25668587
-
Surface modification and endothelialization of biomaterials as potential scaffolds for vascular tissue engineering applications.Chem Soc Rev. 2015 Aug 7;44(15):5680-742. doi: 10.1039/c4cs00483c. Epub 2015 May 29. Chem Soc Rev. 2015. PMID: 26023741 Review.
-
Bioresponsive functional nanogels as an emerging platform for cancer therapy.Expert Opin Drug Deliv. 2018 Jul;15(7):703-716. doi: 10.1080/17425247.2018.1497607. Epub 2018 Jul 16. Expert Opin Drug Deliv. 2018. PMID: 29976103 Review.
Cited by
-
The Collapse of Brain Clearance: Glymphatic-Venous Failure, Aquaporin-4 Breakdown, and AI-Empowered Precision Neurotherapeutics in Intracranial Hypertension.Int J Mol Sci. 2025 Jul 25;26(15):7223. doi: 10.3390/ijms26157223. Int J Mol Sci. 2025. PMID: 40806356 Free PMC article. Review.
-
Biodegradable polymeric occluder with controllable locking structure for closure of atrial septal defect via interventional treatment.Regen Biomater. 2025 Mar 20;12:rbaf016. doi: 10.1093/rb/rbaf016. eCollection 2025. Regen Biomater. 2025. PMID: 40248504 Free PMC article.
References
-
- Zhao J, Feng Y.. Surface engineering of cardiovascular devices for improved hemocompatibility and rapid endothelialization. Adv Healthc Mater 2020;9:e2000920. - PubMed
-
- Jana S. Endothelialization of cardiovascular devices. Acta Biomater 2019;99:53–71. - PubMed
-
- Zheng W, Liu M, Qi H, Wen C, Zhang C, Mi J, Zhou X, Zhang L, Fan D.. Mussel-inspired triblock functional protein coating with endothelial cell selectivity for endothelialization. J Colloid Interface Sci 2020;576:68–78. - PubMed
-
- Khan M, Yang J, Shi C, Lv J, Feng Y, Zhang W.. Surface tailoring for selective endothelialization and platelet inhibition via a combination of SI-ATRP and click chemistry using Cys–Ala–Gly-peptide. Acta Biomater 2015;20:69–81. - PubMed
LinkOut - more resources
Full Text Sources

