Associations of Gestational Exposure to Air Pollution and Polycyclic Aromatic Hydrocarbons with Placental Inflammation
- PMID: 39323894
- PMCID: PMC11420950
- DOI: 10.1021/envhealth.4c00077
Associations of Gestational Exposure to Air Pollution and Polycyclic Aromatic Hydrocarbons with Placental Inflammation
Abstract
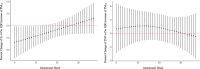
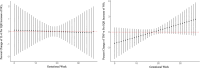
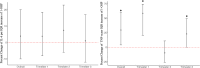
Restricted fetal growth (RFG) is a leading contributor to perinatal mortality and has been associated with gestational exposure to air pollution, such as fine particulate matter (PM2.5), nitrogen dioxide (NO2), and polycyclic aromatic hydrocarbons (PAHs). This study examines the association between trimester-specific and weekly means of air pollution throughout gestation and placental inflammatory markers at delivery. In a prospective cohort study of 263 pregnant women in Rochester, NY, we measured interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) in placental tissue and estimated gestational exposure to PM2.5 and NO2 using a high-resolution spatial-temporal model. Exposure to PAHs was estimated using urinary 1-hydroxypyrene (1-OHP) concentrations collected once per trimester. Using distributed lag models with a penalized spline function, each interquartile range (2.6 μg/m3) increase in PM2.5 concentration during gestational weeks 6-11 was associated with decreased placental IL-6 levels (-22.2%, 95% CI: -39.0%, -0.64%). Using multiple linear regression models, each interquartile range increase of 1-OHP was associated with an increase in TNF-α in the first trimester (58.5%, 95% CI: 20.7%, 74.2%), third trimester (22.9%, 95% CI: 0.04%, 49.5%), and entire pregnancy (29.6%, 95%CI: 3.9%,60.6%). Our results suggest gestational exposure to air pollution may alter the inflammatory environment of the placenta at delivery.
© 2024 The Authors. Co-published by Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, and American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Systemic oxidative stress levels during the course of pregnancy: Associations with exposure to air pollutants.Environ Pollut. 2024 Sep 15;357:124463. doi: 10.1016/j.envpol.2024.124463. Epub 2024 Jun 26. Environ Pollut. 2024. PMID: 38942277
-
Ambient air pollution exposure during the late gestational period is linked with lower placental iodine load in a Belgian birth cohort.Environ Int. 2021 Feb;147:106334. doi: 10.1016/j.envint.2020.106334. Epub 2020 Dec 24. Environ Int. 2021. PMID: 33360673 Free PMC article.
-
Mortality and Morbidity Effects of Long-Term Exposure to Low-Level PM2.5, BC, NO2, and O3: An Analysis of European Cohorts in the ELAPSE Project.Res Rep Health Eff Inst. 2021 Sep;2021(208):1-127. Res Rep Health Eff Inst. 2021. PMID: 36106702 Free PMC article.
-
Mortality-Air Pollution Associations in Low Exposure Environments (MAPLE): Phase 2.Res Rep Health Eff Inst. 2022 Jul;2022(212):1-91. Res Rep Health Eff Inst. 2022. PMID: 36224709 Free PMC article.
-
Assessing Adverse Health Effects of Long-Term Exposure to Low Levels of Ambient Air Pollution: Implementation of Causal Inference Methods.Res Rep Health Eff Inst. 2022 Jan;2022(211):1-56. Res Rep Health Eff Inst. 2022. PMID: 36193708 Free PMC article. Review.
References
-
- Kamphof H. D.; Posthuma S.; Gordijn S. J.; Ganzevoort W. Fetal Growth Restriction: Mechanisms, Epidemiology, and Management. Matern.-Fetal Med. 2022, 4 (3), 186.10.1097/FM9.0000000000000161. - DOI
-
- Rich D. Q.; Liu K.; Zhang J.; Thurston S. W.; Stevens T. P.; Pan Y.; Kane C.; Weinberger B.; Ohman-Strickland P.; Woodruff T. J.; Duan X.; Assibey-Mensah V.; Zhang J. Differences in Birth Weight Associated with the 2008 Beijing Olympics Air Pollution Reduction: Results from a Natural Experiment. Environ. Health Perspect. 2015, 123 (9), 880–887. 10.1289/ehp.1408795. - DOI - PMC - PubMed
-
- Shao X.; Cheng H.; Zhou J.; Zhang J.; Zhu Y.; Yang C.; Di Narzo A.; Yu J.; Shen Y.; Li Y.; Xu S.; Zhang Z.; Chen J.; Cheng J.; Hao K. Prenatal Exposure to Ambient Air Multi-Pollutants Significantly Impairs Intrauterine Fetal Development Trajectory. Ecotoxicol. Environ. Saf. 2020, 201, 110726.10.1016/j.ecoenv.2020.110726. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
