Biospecimens in the HEALthy Brain and Child Development (HBCD) Study: Rationale and protocol
- PMID: 39326174
- PMCID: PMC11460495
- DOI: 10.1016/j.dcn.2024.101451
Biospecimens in the HEALthy Brain and Child Development (HBCD) Study: Rationale and protocol
Abstract
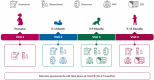
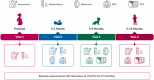
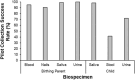
The HEALthy Brain and Child Development (HBCD) Study, a multi-site prospective longitudinal cohort study, will examine human brain, cognitive, behavioral, social, and emotional development beginning prenatally and planned through early childhood. The longitudinal collection of biological samples from over 7000 birthing parents and their children within the HBCD study enables research on pre- and postnatal exposures (e.g., substance use, toxicants, nutrition), and biological processes (e.g., genetics, epigenetic signatures, proteins, metabolites) on neurobehavioral developmental outcomes. The following biosamples are collected from the birthing parent: 1) blood (i.e., whole blood, serum, plasma, buffy coat, and dried blood spots) during pregnancy, 2) nail clippings during pregnancy and one month postpartum, 3) urine during pregnancy, and 4) saliva during pregnancy and at in-person postnatal assessments. The following samples are collected from the child at in-person study assessments: 1) saliva, 2) stool, and 3) urine. Additionally, placenta tissue, cord blood, and cord tissue are collected by a subset of HBCD sites. Here, we describe the rationale for the collection of these biospecimens, their current and potential future uses, the collection protocol, and collection success rates during piloting. This information will assist research teams in the planning of future studies utilizing this collection of biological samples.
Keywords: Environmental exposures; HBCD, biospecimens, substance use; Prenatal, fetal programming.
Copyright © 2024 The Authors. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Drs. Elinor L Sullivan, Ryan Bogdan, Ludmila Bakhireva, Pat Levitt, Julie M. Croff, Moriah Thomason(,) Jamie Lo(,) Leigh-Anne Cioffredi, Andrea G. Edlow, Brittany R. Howell, Barbara H Chaiyachati, Namasivayam Ambalavanan(,) and Jenae M. Neiderhiser report financial support provided by National Institutes of Health that includes funding grants. In addition, the Children's Hospital of Philadelphia has received payment for the expert testimony of Dr. Chaiyachati when subpoenaed for cases of suspected child abuse. Andrea G. Edlow serves as a consultant for Mirvie, Inc. outside of this work and receives research funding and consultant funding from Merck Pharmaceuticals outside of this work. Julie M. Croff reports financial support provided by Bill & Melinda Gates Foundation and reports a relationship with Oklahoma State University Center for Health Sciences that includes: funding grants, speaking and lecture fees, and travel reimbursement. Joseph Jones is employed by United States Drug Testing Laboratories, a national commercial reference laboratory, that is in the business of selling some of the services mentioned in this manuscript. This relationship did not influence the design or composition of this manuscript. Leigh MacIntyre discloses the following financial interests that may be considered as potential competing interests: She is the CEO and founder of Lasso Informatics, and has an equity stake and receives a salary from the company. Lasso Informatics is involved in the HBCD Study and provides project management and data services. During the course of this research, Lasso Informatics received a professional services contract from HBCD, providing financial compensation for Lasso Informatics’ contribution to the study. This contract was awarded in 2024 and pertains to the work reported in this manuscript. Leigh MacIntyre declares that these financial interests have been disclosed fully and believe they do not compromise the integrity of the research presented in this manuscript. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
References
-
- AAFS Standards Board, 2019. Standard Practices for Method Validation in Forensic Toxicology.
-
- Bakhireva L.N., Nebeker C., Ossorio P., Angal J., Thomason M.E., Croff J.M. Inclusion of American Indians and Alaskan Natives in Large National Studies: Ethical Considerations and Implications for Biospecimen Collection in the HEALthy Brain and Child Development Study. Advers Resil. Sci. 2020;1:285–294. doi: 10.1007/s42844-020-00020-8. - DOI - PMC - PubMed
-
- Baranger D.A.A., Miller A.P., Gorelik A.J., Paul S.E., Hatoum A.S., Johnson E.C., Colbert S.M.C., Smyser C.D., Rogers C.E., Bijsterbosch J.D., Agrawal A., Bogdan R. Prenatal cannabis exposure, the brain, and psychopathology during early adolescence. Nat. Ment. Health 1–12. 2024 doi: 10.1038/s44220-024-00281-7. - DOI
Publication types
MeSH terms
Grants and funding
- U01 DA055370/DA/NIDA NIH HHS/United States
- U01 DA055355/DA/NIDA NIH HHS/United States
- U01 DA055361/DA/NIDA NIH HHS/United States
- U01 DA055338/DA/NIDA NIH HHS/United States
- U01 DA055344/DA/NIDA NIH HHS/United States
- U01 DA055316/DA/NIDA NIH HHS/United States
- U01 DA055349/DA/NIDA NIH HHS/United States
- U01 DA055359/DA/NIDA NIH HHS/United States
- U01 DA055354/DA/NIDA NIH HHS/United States
- U01 DA055369/DA/NIDA NIH HHS/United States
- U01 DA055366/DA/NIDA NIH HHS/United States
- U01 DA055357/DA/NIDA NIH HHS/United States
- U01 DA055360/DA/NIDA NIH HHS/United States
- U01 DA055350/DA/NIDA NIH HHS/United States
- U01 DA055347/DA/NIDA NIH HHS/United States
- U01 DA055367/DA/NIDA NIH HHS/United States
- U54 GM115516/GM/NIGMS NIH HHS/United States
- UL1 TR002369/TR/NCATS NIH HHS/United States
- R01 HD113188/HD/NICHD NIH HHS/United States
- U24 DA055330/DA/NIDA NIH HHS/United States
- U01 DA055371/DA/NIDA NIH HHS/United States
- U01 DA055342/DA/NIDA NIH HHS/United States
- U24 DA055325/DA/NIDA NIH HHS/United States
- U01 DA055363/DA/NIDA NIH HHS/United States
- U01 DA055322/DA/NIDA NIH HHS/United States
- U01 DA055353/DA/NIDA NIH HHS/United States
- U01 DA055358/DA/NIDA NIH HHS/United States
- U01 DA055365/DA/NIDA NIH HHS/United States
- U01 DA055362/DA/NIDA NIH HHS/United States

