Surufatinib plus toripalimab combined with etoposide and cisplatin as first-line treatment in advanced small-cell lung cancer patients: a phase Ib/II trial
- PMID: 39327433
- PMCID: PMC11427686
- DOI: 10.1038/s41392-024-01974-2
Surufatinib plus toripalimab combined with etoposide and cisplatin as first-line treatment in advanced small-cell lung cancer patients: a phase Ib/II trial
Abstract
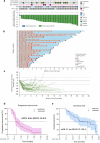
There is still room for improvement in first-line treatment of advanced small cell lung cancer (SCLC). This trial firstly investigated efficacy and safety of antiangiogenic therapy (surufatinib) (200 mg, qd, po) plus anti-PD-1 treatment (toripalimab) (240 mg, d1, ivdrip) combined with etoposide (100 mg/m², d1-d3, iv, drip) and cisplatin (25 mg/m², d1-d3, ivdrip) for advanced SCLC as first-line treatment, which has been registered on ClinicalTrials.gov under the identifier NCT04996771. The four-drug regimen was conducted q3w for 4 cycles with maintenance therapy of surufatinib and toripalimab. The primary endpoint was progression-free survival (PFS). The secondary end points included objective response rate (ORR), disease control rate (DCR), overall survival (OS) and safety. All of the 38 patients were enrolled for safety analysis, while only 35 patients were enrolled for efficacy analysis since loss of efficacy evaluation in 3 cases after treatment. After a median follow-up of 21.3 months, the ORR was 97.1% (34/35), and the DCR and the tumor shrinkage rate were both 100% (35/35). The median PFS was 6.9 months (95% CI: 4.6 m-9.2 m) and the median OS was 21.1 months (95% CI: 12.1 m-30.1 m). The 12-month, 18-month, and 24-month OS rates were 66.94%, 51.39% and 38.54%. The occurrence rate of grade ≥3 treatment-emergent adverse events (TEAEs) was 63.2% (24/38), including neutrophil count decreased (31.6%, 12/38), white blood cell count decreased (23.7%, 9/38) and platelet count decreased (10.5%, 4/38). No unexpected adverse events occurred. This novel four-drug regimen (surufatinib, toripalimab, etoposide plus cisplatin) revealed impressive therapeutic efficacy and tolerable toxicities.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Toripalimab Plus Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Lung Cancer: The Phase 3 EXTENTORCH Randomized Clinical Trial.JAMA Oncol. 2025 Jan 1;11(1):16-25. doi: 10.1001/jamaoncol.2024.5019. JAMA Oncol. 2025. PMID: 39541202 Clinical Trial.
-
Efficacy and safety of surufatinib plus toripalimab in treatment-naive, PD-L1-positive, advanced or metastatic non-small-cell lung cancer and previously treated small-cell lung cancer: an open-label, single-arm, multicenter, multi-cohort phase II trial.Cancer Immunol Immunother. 2025 Feb 1;74(3):83. doi: 10.1007/s00262-024-03932-x. Cancer Immunol Immunother. 2025. PMID: 39891720 Free PMC article. Clinical Trial.
-
Zanidatamab plus chemotherapy as first-line treatment for patients with HER2-positive advanced gastro-oesophageal adenocarcinoma: primary results of a multicentre, single-arm, phase 2 study.Lancet Oncol. 2025 Jul;26(7):847-859. doi: 10.1016/S1470-2045(25)00287-6. Epub 2025 Jun 2. Lancet Oncol. 2025. PMID: 40473445 Clinical Trial.
-
Comparison of first-line chemotherapy including escalated BEACOPP versus chemotherapy including ABVD for people with early unfavourable or advanced stage Hodgkin lymphoma.Cochrane Database Syst Rev. 2017 May 25;5(5):CD007941. doi: 10.1002/14651858.CD007941.pub3. Cochrane Database Syst Rev. 2017. PMID: 28541603 Free PMC article.
-
Comparison of efficacy and safety of PD-1/PD-L1 combination therapy in first-line treatment of advanced NSCLC: an updated systematic review and network meta-analysis.Clin Transl Oncol. 2024 Oct;26(10):2488-2502. doi: 10.1007/s12094-024-03442-3. Epub 2024 Apr 16. Clin Transl Oncol. 2024. PMID: 38625495
Cited by
-
Predictors of febrile neutropenia in small cell lung cancer patients receiving concurrent chemoradiotherapy with etoposide and cisplatin: a focus on nutritional status, inflammation, and performance status.Am J Cancer Res. 2025 Mar 15;15(3):1020-1035. doi: 10.62347/JRMG1142. eCollection 2025. Am J Cancer Res. 2025. PMID: 40226480 Free PMC article.
-
Advances in biomarkers for immunotherapy in small-cell lung cancer.Front Immunol. 2024 Dec 11;15:1490590. doi: 10.3389/fimmu.2024.1490590. eCollection 2024. Front Immunol. 2024. PMID: 39723215 Free PMC article. Review.
-
The adverse events of toripalimab in nasopharyngeal carcinoma based on FAERS database and bibliometric analysis.PLoS One. 2025 Jun 20;20(6):e0326216. doi: 10.1371/journal.pone.0326216. eCollection 2025. PLoS One. 2025. PMID: 40540517 Free PMC article.
-
Insights into CSF-1/CSF-1R signaling: the role of macrophage in radiotherapy.Front Immunol. 2025 Feb 3;16:1530890. doi: 10.3389/fimmu.2025.1530890. eCollection 2025. Front Immunol. 2025. PMID: 40007537 Free PMC article. Review.
-
Induction chemotherapy followed by camrelizumab plus apatinib and chemotherapy as first-line treatment for extensive-stage small-cell lung cancer: a multicenter, single-arm trial.Signal Transduct Target Ther. 2025 Feb 18;10(1):65. doi: 10.1038/s41392-025-02153-7. Signal Transduct Target Ther. 2025. PMID: 39962074 Free PMC article. Clinical Trial.
References
-
- International Agency for Research on Cancer World Health Organization. GLOBOCAN. http://globocan.iarc.fr.
-
- Key Statistics for Lung Cancer. Atlanta GACS. American Cancer Society. https://www.cancer.org/cancer/lung-cancer/about/key-statistics.html.
-
- Gazdar, A. F., Bunn, P. A. & Minna, J. D. Small-cell lung cancer: what we know, what we need to know and the path forward. Nat. Rev. Cancer17, 725–737 (2017). - PubMed
-
- Rudin, C. M. et al. Treatment of small-cell lung cancer: American Society of Clinical Oncology Endorsement of the American College of Chest Physicians Guideline. J. Clin. Oncol.33, 4106–4111 (2015). - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

