Metabolic Reprogramming in Glioblastoma Multiforme: A Review of Pathways and Therapeutic Targets
- PMID: 39329757
- PMCID: PMC11430559
- DOI: 10.3390/cells13181574
Metabolic Reprogramming in Glioblastoma Multiforme: A Review of Pathways and Therapeutic Targets
Abstract
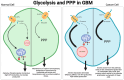
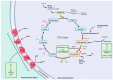
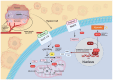
Glioblastoma (GBM) is an aggressive and highly malignant primary brain tumor characterized by rapid growth and a poor prognosis for patients. Despite advancements in treatment, the median survival time for GBM patients remains low. One of the crucial challenges in understanding and treating GBMs involves its remarkable cellular heterogeneity and adaptability. Central to the survival and proliferation of GBM cells is their ability to undergo metabolic reprogramming. Metabolic reprogramming is a process that allows cancer cells to alter their metabolism to meet the increased demands of rapid growth and to survive in the often oxygen- and nutrient-deficient tumor microenvironment. These changes in metabolism include the Warburg effect, alterations in several key metabolic pathways including glutamine metabolism, fatty acid synthesis, and the tricarboxylic acid (TCA) cycle, increased uptake and utilization of glutamine, and more. Despite the complexity and adaptability of GBM metabolism, a deeper understanding of its metabolic reprogramming offers hope for developing more effective therapeutic interventions against GBMs.
Keywords: Warburg effect; glioblastoma multiforme; glycolysis; metabolic reprogramming; therapeutic drugs; tumor microenvironment.
Conflict of interest statement
Betty Tyler has research funding from NIH. Ashvattha Therapeutics Inc.* has licensed one of her patents, and she is a stockholder for Peabody Pharmaceuticals* (*includes equity or options).
Figures
Similar articles
-
Metabolic reprogramming in glioblastoma: the influence of cancer metabolism on epigenetics and unanswered questions.Neuro Oncol. 2016 Feb;18(2):160-72. doi: 10.1093/neuonc/nov125. Epub 2015 Jul 14. Neuro Oncol. 2016. PMID: 26180081 Free PMC article. Review.
-
Beyond glucose: alternative sources of energy in glioblastoma.Theranostics. 2021 Jan 1;11(5):2048-2057. doi: 10.7150/thno.53506. eCollection 2021. Theranostics. 2021. PMID: 33500708 Free PMC article. Review.
-
The Impact of Metabolic Rewiring in Glioblastoma: The Immune Landscape and Therapeutic Strategies.Int J Mol Sci. 2025 Jan 14;26(2):669. doi: 10.3390/ijms26020669. Int J Mol Sci. 2025. PMID: 39859381 Free PMC article. Review.
-
Glioma cells require one-carbon metabolism to survive glutamine starvation.Acta Neuropathol Commun. 2021 Jan 19;9(1):16. doi: 10.1186/s40478-020-01114-1. Acta Neuropathol Commun. 2021. PMID: 33468252 Free PMC article.
-
Metabolic shifts in glioblastoma: unraveling altered pathways and exploring novel therapeutic avenues.Mol Biol Rep. 2025 Jan 22;52(1):146. doi: 10.1007/s11033-025-10242-7. Mol Biol Rep. 2025. PMID: 39841290 Review.
Cited by
-
High-Throughput Targeted Drug Screening for NF1-associated High-Grade Gliomas with ATRX Deficiency.bioRxiv [Preprint]. 2025 Jun 11:2024.12.14.628525. doi: 10.1101/2024.12.14.628525. bioRxiv. 2025. PMID: 40661612 Free PMC article. Preprint.
-
APOL4-mediated intracellular cholesterol trafficking is essential for glioblastoma cell growth.BMC Cancer. 2025 May 21;25(1):906. doi: 10.1186/s12885-025-14316-4. BMC Cancer. 2025. PMID: 40399861 Free PMC article.
-
Fumonisin B1 induces global DNA hypermethylation in human glioblastoma U87MG cells.Epigenetics. 2025 Dec;20(1):2523690. doi: 10.1080/15592294.2025.2523690. Epub 2025 Jun 26. Epigenetics. 2025. PMID: 40566980 Free PMC article.
-
Spatial tumour characteristics as an indirect marker of metabolic dysregulation: evaluation for non-invasive IDH-genotyping of glioma using hybrid [18 F]FET-PET/MRI.Eur J Nucl Med Mol Imaging. 2025 Aug 28. doi: 10.1007/s00259-025-07520-8. Online ahead of print. Eur J Nucl Med Mol Imaging. 2025. PMID: 40866645
-
Identification of Prognostic Genes Related to Cell Senescence and Lipid Metabolism in Glioblastoma Based on Transcriptome and Single-Cell RNA-Seq Data.Int J Mol Sci. 2025 Feb 21;26(5):1875. doi: 10.3390/ijms26051875. Int J Mol Sci. 2025. PMID: 40076502 Free PMC article.
References
-
- Rock K., McArdle O., Forde P., Dunne M., Fitzpatrick D., O’Neill B., Faul C. A clinical review of treatment outcomes in glioblastoma multiforme—The validation in a non-trial population of the results of a randomised Phase III clinical trial: Has a more radical approach improved survival? Br. J. Radiol. 2012;85:e729–e733. doi: 10.1259/bjr/83796755. - DOI - PMC - PubMed
-
- Trejo-Solis C., Silva-Adaya D., Serrano-García N., Magaña-Maldonado R., Jimenez-Farfan D., Ferreira-Guerrero E., Cruz-Salgado A., Castillo-Rodriguez R.A. Role of Glycolytic and Glutamine Metabolism Reprogramming on the Proliferation, Invasion, and Apoptosis Resistance through Modulation of Signaling Pathways in Glioblastoma. Int. J. Mol. Sci. 2023;24:17633. doi: 10.3390/ijms242417633. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

