NOD1 and NOD2: Essential Monitoring Partners in the Innate Immune System
- PMID: 39329913
- PMCID: PMC11430502
- DOI: 10.3390/cimb46090561
NOD1 and NOD2: Essential Monitoring Partners in the Innate Immune System
Abstract
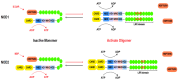
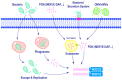
Nucleotide-binding oligomerization domain containing 1 (NOD1) and NOD2 are pivotal cytoplasmic pattern-recognition receptors (PRRs) that exhibit remarkable evolutionary conservation. They possess the ability to discern specific peptidoglycan (PGN) motifs, thereby orchestrating innate immunity and contributing significantly to immune homeostasis maintenance. The comprehensive understanding of both the structure and function of NOD1 and NOD2 has been extensively elucidated. These receptors proficiently recognize an array of damage-associated molecular patterns (DAMPs) as well as pathogen-associated molecular patterns (PAMPs), subsequently mediating inflammatory responses and autophagy. In recent years, emerging evidence has highlighted the crucial roles played by NOD1 and NOD2 in regulating infectious diseases, metabolic disorders, cancer, and autoimmune conditions, among others. Perturbation in either their loss or excessive activation can detrimentally impact immune homeostasis. This review offers a comprehensive overview of the structural characteristics, subcellular localization, activation mechanisms, and significant roles of NOD1 and NOD2 in innate immunity and related disease.
Keywords: NOD1; NOD2; inflammatory response; innate immunity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
NOD1 and NOD2 Are Potential Therapeutic Targets for Cancer Immunotherapy.Comput Intell Neurosci. 2022 Oct 10;2022:2271788. doi: 10.1155/2022/2271788. eCollection 2022. Comput Intell Neurosci. 2022. Retraction in: Comput Intell Neurosci. 2023 Aug 9;2023:9847813. doi: 10.1155/2023/9847813. PMID: 36262606 Free PMC article. Retracted. Review.
-
NOD1 and NOD2: signaling, host defense, and inflammatory disease.Immunity. 2014 Dec 18;41(6):898-908. doi: 10.1016/j.immuni.2014.12.010. Epub 2014 Dec 6. Immunity. 2014. PMID: 25526305 Free PMC article. Review.
-
NOD1 and NOD2: Molecular targets in prevention and treatment of infectious diseases.Int Immunopharmacol. 2018 Jan;54:385-400. doi: 10.1016/j.intimp.2017.11.036. Epub 2017 Dec 5. Int Immunopharmacol. 2018. PMID: 29207344 Review.
-
Structural models of zebrafish (Danio rerio) NOD1 and NOD2 NACHT domains suggest differential ATP binding orientations: insights from computational modeling, docking and molecular dynamics simulations.PLoS One. 2015 Mar 26;10(3):e0121415. doi: 10.1371/journal.pone.0121415. eCollection 2015. PLoS One. 2015. PMID: 25811192 Free PMC article.
-
Roles of NOD1 (NLRC1) and NOD2 (NLRC2) in innate immunity and inflammatory diseases.Biosci Rep. 2012 Dec;32(6):597-608. doi: 10.1042/BSR20120055. Biosci Rep. 2012. PMID: 22908883 Free PMC article. Review.
Cited by
-
Molecular Identification and Expression Analysis of NOD1/2 and TBK1 in Response to Viral or Bacterial Infection in the Spotted Knifejaw (Oplegnathus punctatus).Animals (Basel). 2025 Mar 31;15(7):1006. doi: 10.3390/ani15071006. Animals (Basel). 2025. PMID: 40218399 Free PMC article.
-
Effects of rumen-protected lysine on antler growth performance, fecal bacterial community, and blood gene expression in sika deer.Front Vet Sci. 2025 Jul 11;12:1583605. doi: 10.3389/fvets.2025.1583605. eCollection 2025. Front Vet Sci. 2025. PMID: 40717913 Free PMC article.
-
κ-carrageenan oligosaccharides alleviate MDP induced rumen epithelial cell inflammatory damage by inhibiting the activation of NOD2/NF-κB pathway.Front Vet Sci. 2025 Jul 9;12:1626423. doi: 10.3389/fvets.2025.1626423. eCollection 2025. Front Vet Sci. 2025. PMID: 40703925 Free PMC article.
-
The known genetic variants of BRCA1, BRCA2 and NOD2 in pancreatitis and pancreatic cancer risk assessment.Sci Rep. 2025 Jan 13;15(1):1791. doi: 10.1038/s41598-025-86249-8. Sci Rep. 2025. PMID: 39805914 Free PMC article.
-
NOD2 Polymorphisms and Their Association with Colorectal Cancer Risk: An Updated Systematic Review and Meta-Analysis.Cancers (Basel). 2025 Jun 15;17(12):1999. doi: 10.3390/cancers17121999. Cancers (Basel). 2025. PMID: 40563649 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

