The Impact of the Methacrylation Process on the Usefulness of Chitosan as a Biomaterial Component for 3D Printing
- PMID: 39330227
- PMCID: PMC11433516
- DOI: 10.3390/jfb15090251
The Impact of the Methacrylation Process on the Usefulness of Chitosan as a Biomaterial Component for 3D Printing
Abstract
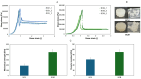
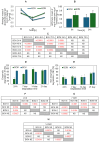
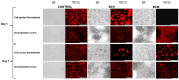
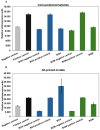
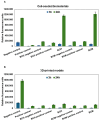
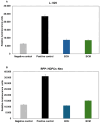
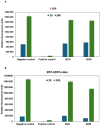
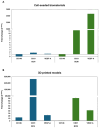
Chitosan is a very promising material for tissue model printing. It is also known that the introduction of chemical modifications to the structure of the material in the form of methacrylate groups makes it very attractive for application in the bioprinting of tissue models. The aim of this work is to study the characteristics of biomaterials containing chitosan (BCH) and its methacrylated equivalent (BCM) in order to identify differences in their usefulness in 3D bioprinting technology. It has been shown that the BCM material containing methacrylic chitosan is three times more viscous than its non-methacrylated BCH counterpart. Additionally, the BCM material is characterized by stability in a larger range of stresses, as well as better printability, resolution, and fiber stability. The BCM material has higher mechanical parameters, both mechanical strength and Young's modulus, than the BCH material. Both materials are ideal for bioprinting, but BCM has unique rheological properties and significant mechanical resistance. In addition, biological tests have shown that the addition of chitosan to biomaterials increases cell proliferation, particularly in 3D-printed models. Moreover, modification in the form of methacrylation encourages reduced toxicity of the biomaterial in 3D constructs. Our investigation demonstrates the suitability of a chitosan-enhanced biomaterial, specifically methacrylate-treated, for application in tissue engineering, and particularly for tissues requiring resistance to high stress, i.e., vascular or cartilage models.
Keywords: biomaterial; bioprinting; chitosan; extracellular matrix; methacrylation; tissue engineering; tissue regeneration.
Conflict of interest statement
Authors Marta Klak, Katarzyna Kosowska, Milena Czajka, Magdalena Dec, Sylwester Domański, Agnieszka Zakrzewska, and Michał Wszoła are employed by the company Polbionica Ltd. The other authors declare no conflicts of interest.
Figures


Similar articles
-
Three-Dimensional-Printable Thermo/Photo-Cross-Linked Methacrylated Chitosan-Gelatin Hydrogel Composites for Tissue Engineering.ACS Appl Mater Interfaces. 2021 May 19;13(19):22902-22913. doi: 10.1021/acsami.1c01321. Epub 2021 May 7. ACS Appl Mater Interfaces. 2021. PMID: 33960765
-
Bioprinting of alginate-carboxymethyl chitosan scaffolds for enamel tissue engineeringin vitro.Biofabrication. 2022 Dec 30;15(1). doi: 10.1088/1758-5090/acab35. Biofabrication. 2022. PMID: 36583240
-
A rheological approach to assess the printability of thermosensitive chitosan-based biomaterial inks.Biomed Mater. 2020 Nov 27;16(1):015003. doi: 10.1088/1748-605X/abb2d8. Biomed Mater. 2020. PMID: 33245047
-
Techniques and applications in 3D bioprinting with chitosan bio-inks for drug delivery: A review.Int J Biol Macromol. 2024 Oct;278(Pt 4):134752. doi: 10.1016/j.ijbiomac.2024.134752. Epub 2024 Aug 30. Int J Biol Macromol. 2024. PMID: 39214837 Review.
-
Biomaterials in bone and mineralized tissue engineering using 3D printing and bioprinting technologies.Biomed Phys Eng Express. 2021 Oct 7;7(6). doi: 10.1088/2057-1976/ac21ab. Biomed Phys Eng Express. 2021. PMID: 34438382 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

