Putative Role of Adenosine A1 Receptors in Exogenous Ketone Supplements-Evoked Anti-Epileptic Effect
- PMID: 39337356
- PMCID: PMC11432942
- DOI: 10.3390/ijms25189869
Putative Role of Adenosine A1 Receptors in Exogenous Ketone Supplements-Evoked Anti-Epileptic Effect
Abstract
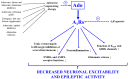
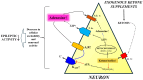
Approximately 30% of patients with epilepsy are drug-refractory. There is an urgent need to elucidate the exact pathophysiology of different types of epilepsies and the mechanisms of action of both antiseizure medication and metabolic therapies to treat patients more effectively and safely. For example, it has been demonstrated that exogenous ketone supplement (EKS)-generated therapeutic ketosis, as a metabolic therapy, may decrease epileptic activity in both animal models and humans, but its exact mechanism of action is unknown. However, it was demonstrated that therapeutic ketosis, among others, can increase adenosine level, which may enhance activity of A1 adenosine receptors (A1Rs) in the brain. It has also been demonstrated previously that adenosine has anti-epileptic effect through A1Rs in different models of epilepsies. Thus, it is possible that (i) therapeutic ketosis generated by the administration of EKSs may exert its anti-epileptic effect through, among other mechanisms, increased adenosine level and A1R activity and that (ii) the enhanced activity of A1Rs may be a necessary anti-epileptic mechanism evoked by EKS administration-generated ketosis. Moreover, EKSs can evoke and maintain ketosis without severe side effects. These results also suggest that the therapeutic application of EKS-generated ketosis may be a promising opportunity to treat different types of epilepsies. In this literature review, we specifically focus on the putative role of A1Rs in the anti-epileptic effect of EKS-induced ketosis.
Keywords: A1R; epilepsy; exogenous ketone supplement; ketosis.
Conflict of interest statement
Patent US10980764B1 C. Ari, D.P. D’Agostino, “Exogenous ketone supplements for reducing anxiety-related behavior”. C. Ari and D.P. D’Agostino are co-owners of Ketone Technologies LLC and C. Ari is owner of Fortis World LLC. C. Ari was employed by Ketone Technologies LLC and Fortis World LLC. These interests have been reviewed and managed by the University in accordance with its Institutional and Individual Conflict of Interest policies. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. All authors declare that there are no additional conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

