Wnt/β-Catenin Signaling Regulates Yap/Taz Activity during Embryonic Development in Zebrafish
- PMID: 39337493
- PMCID: PMC11432159
- DOI: 10.3390/ijms251810005
Wnt/β-Catenin Signaling Regulates Yap/Taz Activity during Embryonic Development in Zebrafish
Abstract
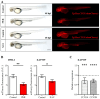
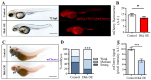
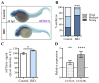
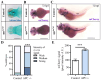
Hippo-YAP/TAZ and Wnt/β-catenin signaling pathways, by controlling proliferation, migration, cell fate, stemness, and apoptosis, are crucial regulators of development and tissue homeostasis. We employed zebrafish embryos as a model system to elucidate in living reporter organisms the crosstalk between the two signaling pathways. Co-expression analysis between the Wnt/β-catenin Tg(7xTCF-Xla.Siam:GFP)ia4 and the Hippo-Yap/Taz Tg(Hsa.CTGF:nlsmCherry)ia49 zebrafish reporter lines revealed shared spatiotemporal expression profiles. These patterns were particularly evident in key developmental regions such as the midbrain-hindbrain boundary (MHB), epidermis, muscles, neural tube, notochord, floorplate, and otic vesicle. To investigate the relationship between the Wnt/β-catenin pathway and Hippo-Yap/Taz signaling in vivo, we conducted a series of experiments employing both pharmacological and genetic strategies. Modulation of the Wnt/β-catenin pathway with IWR-1, XAV939, or BIO resulted in a significant regulation of the Yap/Taz reporter signal, highlighting a clear correlation between β-catenin and Yap/Taz activities. Furthermore, genetic perturbation of the Wnt/β-catenin pathway, by APC inhibition or DKK1 upregulation, elicited evident and robust alteration of Yap/Taz activity. These findings revealed the intricate regulatory mechanisms underlying the crosstalk between the Wnt/β-catenin and Hippo-Yap/Taz signaling, shedding light on their roles in orchestrating developmental processes in vivo.
Keywords: Hippo; Wnt/β-catenin; Yap/Taz; crosstalk; embryonic development; zebrafish.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
The effects of YAP/TAZ in cardiomyocytes: a scoping review.Mol Biol Rep. 2025 Apr 15;52(1):392. doi: 10.1007/s11033-025-10492-5. Mol Biol Rep. 2025. PMID: 40232357
-
Establishment of transgenic lines to monitor and manipulate Yap/Taz-Tead activity in zebrafish reveals both evolutionarily conserved and divergent functions of the Hippo pathway.Mech Dev. 2014 Aug;133:177-88. doi: 10.1016/j.mod.2014.02.003. Epub 2014 Feb 19. Mech Dev. 2014. PMID: 24560909 Free PMC article.
-
YAP/TAZ as master regulators in liver regeneration and disease: insights into mechanisms and therapeutic targets.Mol Biol Rep. 2024 Dec 24;52(1):78. doi: 10.1007/s11033-024-10177-5. Mol Biol Rep. 2024. PMID: 39718664 Review.
-
Hierarchy of TGFβ/SMAD, Hippo/YAP/TAZ, and Wnt/β-catenin signaling in melanoma phenotype switching.Life Sci Alliance. 2021 Nov 24;5(2):e202101010. doi: 10.26508/lsa.202101010. Print 2022 Feb. Life Sci Alliance. 2021. PMID: 34819356 Free PMC article.
-
Yap and Taz regulate retinal pigment epithelial cell fate.Development. 2015 Sep 1;142(17):3021-32. doi: 10.1242/dev.119008. Epub 2015 Jul 24. Development. 2015. PMID: 26209646 Free PMC article.
Cited by
-
COPB2 promotes hepatocellular carcinoma progression through regulation of YAP1 nuclear translocation.Oncol Res. 2025 Mar 19;33(4):975-988. doi: 10.32604/or.2025.058085. eCollection 2025. Oncol Res. 2025. PMID: 40191726 Free PMC article.
-
Fish-derived biomaterials for tissue engineering: advances in scaffold fabrication and applications in regenerative medicine and cancer therapy.Theranostics. 2025 Apr 21;15(12):5666-5692. doi: 10.7150/thno.109186. eCollection 2025. Theranostics. 2025. PMID: 40365274 Free PMC article. Review.
-
Tuina promotes nerve myelin regeneration in SNI rats through Piezo1/YAP/TAZ pathway.J Orthop Surg Res. 2025 May 12;20(1):454. doi: 10.1186/s13018-025-05794-0. J Orthop Surg Res. 2025. PMID: 40350445 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

