Outer Membrane Vesicle Production by Escherichia coli Enhances Its Defense against Phage Infection
- PMID: 39338510
- PMCID: PMC11433858
- DOI: 10.3390/microorganisms12091836
Outer Membrane Vesicle Production by Escherichia coli Enhances Its Defense against Phage Infection
Abstract
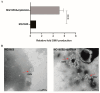
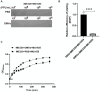
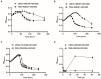
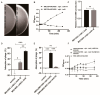
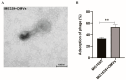
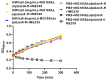
Several studies have investigated the multifunctional characteristics of outer membrane vesicles (OMVs), but research on their role in mediating phage-bacteria interactions is limited. Employing Escherichia coli as a model, we engineered a mutant strain overproducing OMVs for protective experiments against phage infections. The addition of exogenous OMVs proved highly effective in safeguarding the bacterial host against various phages, mitigating predatory threats. Screening for phage-resistant strains and adsorption experiments revealed that inhibiting phage adsorption is a crucial pathway through which OMVs protect against phage predation. Although OMVs conferred tolerance to the phage-sensitive strains (those easily infected by phages), they could not restore the phage-resistant strains (those that effectively resist phage infection) to a sensitive phenotype. This study provides valuable insights for the future development of novel biotechnological approaches aimed at utilizing OMVs to protect fermentative strains and reduce the risk of phage contamination.
Keywords: E. coli; adsorption; outer membrane vesicles; phage; resistance.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Outer Membrane Vesicles (OMVs) of Pseudomonas aeruginosa Provide Passive Resistance but Not Sensitization to LPS-Specific Phages.Viruses. 2022 Jan 11;14(1):121. doi: 10.3390/v14010121. Viruses. 2022. PMID: 35062325 Free PMC article.
-
Outer Membrane Vesicles Protect Gram-Negative Bacteria against Host Defense Peptides.mSphere. 2021 Aug 25;6(4):e0052321. doi: 10.1128/mSphere.00523-21. Epub 2021 Jul 7. mSphere. 2021. PMID: 34232080 Free PMC article.
-
Vibrio cholerae Outer Membrane Vesicles Inhibit Bacteriophage Infection.J Bacteriol. 2018 Jul 10;200(15):e00792-17. doi: 10.1128/JB.00792-17. Print 2018 Aug 1. J Bacteriol. 2018. PMID: 29661863 Free PMC article.
-
Bacterial membrane vesicles: formation, functions, and roles in bacterial-phage interactions.World J Microbiol Biotechnol. 2024 Sep 21;40(10):329. doi: 10.1007/s11274-024-04148-y. World J Microbiol Biotechnol. 2024. PMID: 39304539 Review.
-
Secretion and Delivery of Intestinal Pathogenic Escherichia coli Virulence Factors via Outer Membrane Vesicles.Front Cell Infect Microbiol. 2020 Mar 6;10:91. doi: 10.3389/fcimb.2020.00091. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32211344 Free PMC article. Review.
Cited by
-
[Isolation and proteomic analysis of bacterial outer membrane vesicle subpopulations].Se Pu. 2025 May;43(5):529-538. doi: 10.3724/SP.J.1123.2024.10028. Se Pu. 2025. PMID: 40331616 Free PMC article. Chinese.
References
-
- Cui C., He Q., Wang J., Kang J., Ma W., Nian Y., Sun Z., Weng H. Targeted miR-34a delivery with PD1 displayed bacterial outer membrane vesicles-coated zeolitic imidazolate framework nanoparticles for enhanced tumor therapy. Int. J. Biol. Macromol. 2023;247:125692. doi: 10.1016/j.ijbiomac.2023.125692. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

