Liposomal Drug Delivery against Helicobacter pylori Using Furazolidone and N-Acetyl Cysteine in Augmented Therapy
- PMID: 39339161
- PMCID: PMC11435436
- DOI: 10.3390/pharmaceutics16091123
Liposomal Drug Delivery against Helicobacter pylori Using Furazolidone and N-Acetyl Cysteine in Augmented Therapy
Abstract
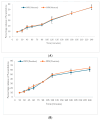
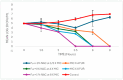
Helicobacter pylori (H. pylori) infection is a significant global health concern, affecting approximately 50% of the world's population and leading to gastric ulcers, gastritis, and gastric cancer. The increase in antibiotic resistance has compromised the efficacy of existing therapeutic regimens, necessitating novel approaches for effective eradication. This study aimed to develop a targeted liposomal drug delivery system incorporating furazolidone and N-acetylcysteine (NAC) to enhance mucopenetration and improve Helicobacter pylori eradication. Liposomes were formulated with furazolidone, NAC, and Pluronic F-127 using a modified reverse-phase evaporation technique. The formulations were categorized based on charge as neutral, negative, and positive and tested for mucopenetration using a modified silicon tube method with coumarin-6 as a fluorescent marker. The encapsulation efficiency and particle size were analyzed using HPLC and an Izon q-nano particle size analyzer. The results indicated that charged liposomes showed a higher encapsulation efficiency than neutral liposomes with Pluronic F-127. Notably, combining furazolidone with 1% NAC achieved complete eradication of H. pylori in 2.5 h, compared to six hours without NAC. The findings of this study suggest that incorporating NAC and Pluronic F-127 into liposomal formulations significantly enhances mucopenetration and antimicrobial efficacy.
Keywords: H. pylori; N-acetyl cysteine; augmented therapy; furazolidone; liposomes; mucus penetration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- FitzGerald R., Smith S.M. An overview of Helicobacter pylori infection. In: Smith S.M., editor. Helicobacter pylori. Volume 2283. Humana; Louisville, KY, USA: 2021. pp. 1–20. - PubMed
-
- Elbehiry A., Marzouk E., Aldubaib M., Abalkhail A., Anagreyyah S., Anajirih N., Almuzaini A.M., Rawway M., Alfadhel A., Draz A., et al. Helicobacter pylori Infection: Current Status and Future Prospects on Diagnostic, Therapeutic and Control Challenges. Antibiotics. 2023;12:191. doi: 10.3390/antibiotics12020191. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

