Comparison of Extraction Methods for the Detection of Avian Influenza Virus RNA in Cattle Milk
- PMID: 39339917
- PMCID: PMC11437502
- DOI: 10.3390/v16091442
Comparison of Extraction Methods for the Detection of Avian Influenza Virus RNA in Cattle Milk
Abstract
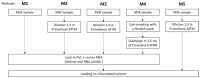
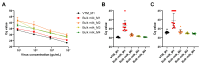
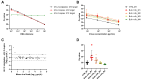
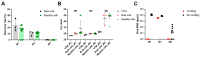
Since early 2024, a multistate outbreak of highly pathogenic avian influenza H5N1 has been affecting dairy cattle in the USA. The influenza viral RNA concentrations in milk make it an ideal matrix for surveillance purposes. However, viral RNA detection in multi-component fluids such as milk can be complex, and optimization of influenza detection methods is thus required. Raw bulk tank milk and mastitis milk samples were artificially contaminated with an avian influenza strain and subjected to five extraction methods. HCoV-229E and synthetic RNA were included as exogenous internal process controls. Given the high viral load usually observed in individual raw milk samples, four out of five tested methods would enable influenza detection in milk with normal texture, over a time window of at least 2 weeks post-onset of clinical signs. Nevertheless, sample dilution 1:3 in molecular transport medium prior to RNA extraction provided the best results for dilution of inhibitory substances and a good recovery rate of influenza RNA, that reached 12.5 ± 1.2% and 10.4 ± 3.8% in two independent experiments in bulk milk and 11.2 ± 3.6% and 10.0 ± 2.9% on two cohorts of mastitis milk samples. We have also shown compatibility of an influenza RT-qPCR system with synthetic RNA detection for simultaneous validation of the RNA extraction and RT-qPCR processes.
Keywords: H5N1; PCR; RNA extraction; dairy cattle; highly pathogenic avian influenza; milk.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Characterization of highly pathogenic avian influenza virus in retail dairy products in the US.J Virol. 2024 Jul 23;98(7):e0088124. doi: 10.1128/jvi.00881-24. Epub 2024 Jul 3. J Virol. 2024. PMID: 38958444 Free PMC article.
-
Comprehensive Insights into Highly Pathogenic Avian Influenza H5N1 in Dairy Cattle: Transmission Dynamics, Milk-Borne Risks, Public Health Implications, Biosecurity Recommendations, and One Health Strategies for Outbreak Control.Pathogens. 2025 Mar 13;14(3):278. doi: 10.3390/pathogens14030278. Pathogens. 2025. PMID: 40137763 Free PMC article. Review.
-
Spillover of highly pathogenic avian influenza H5N1 virus to dairy cattle.Nature. 2024 Oct;634(8034):669-676. doi: 10.1038/s41586-024-07849-4. Epub 2024 Jul 25. Nature. 2024. PMID: 39053575 Free PMC article.
-
Establishing Methods to Monitor Influenza (A)H5N1 Virus in Dairy Cattle Milk, Massachusetts, USA.Emerg Infect Dis. 2025 May;31(13):70-75. doi: 10.3201/eid3113.250087. Epub 2025 Mar 26. Emerg Infect Dis. 2025. PMID: 40138725 Free PMC article.
-
Updated values for molecular diagnosis for highly pathogenic avian influenza virus.Viruses. 2012 Aug;4(8):1235-57. doi: 10.3390/v4081235. Epub 2012 Aug 7. Viruses. 2012. PMID: 23012622 Free PMC article. Review.
References
-
- Nguyen T.-Q., Hutter C., Markin A., Thomas M., Lantz K., Killian M.L., Janzen G.M., Vijendran S., Wagle S., Inderski B., et al. Emergence and interstate spread of highly pathogenic avian influenza A(H5N1) in dairy cattle. bioRxiv. 2024 doi: 10.1101/2024.05.01.591751. - DOI
-
- Caserta L.C., Frye E.A., Butt S.L., Laverack M., Nooruzzaman M., Covaleda L.M., Thompson A.C., Prarat Koscielny M., Cronk B., Johnson A., et al. From birds to mammals: Spillover of highly pathogenic avian influenza H5N1 virus to dairy cattle led to efficient intra- and interspecies transmission. bioRxiv. 2024 doi: 10.1101/2024.05.22.595317. - DOI - PMC - PubMed
-
- USDA Federal and State Veterinary, Public Health Agencies Share Update on HPAI Detection in Kansas, Texas Dairy Herds. [(accessed on 22 July 2024)]; Available online: https://www.aphis.usda.gov/news/agency-announcements/federal-state-veter....
-
- USDA HPAI Confirmed Cases in Livestock. [(accessed on 13 August 2024)]; Available online: https://www.aphis.usda.gov/livestock-poultry-disease/avian/avian-influen....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

