Bactofection, Bacterial-Mediated Vaccination, and Cancer Therapy: Current Applications and Future Perspectives
- PMID: 39340000
- PMCID: PMC11435753
- DOI: 10.3390/vaccines12090968
Bactofection, Bacterial-Mediated Vaccination, and Cancer Therapy: Current Applications and Future Perspectives
Abstract
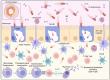
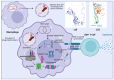
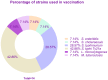
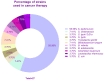
From the first report in 1891 by Dr. Coley of the effective treatment of tumors in 1000 patients with Streptococcus and the first successful use of bacterial vectors for transferring therapeutic genes in 1980 by Dr. Schnaffer, bactofection has been shown to be a promising strategy in the fields of vaccination, gene therapy, and cancer therapy. This review describes the general theory of bactofection and its advantages, disadvantages, challenges, and expectations, compiling the most notable advances in 14 vaccination studies, 27 cancer therapy studies, and 13 clinical trials. It also describes the current scope of bactofection and promising results. The extensive knowledge of Salmonella biology, as well as the multiple adequacies of the Ty21a vaccination platform, has allowed notable developments worldwide that have mainly been reflected in therapeutic efforts against cancer. In this regard, we strongly recommend the creation of a recombinant Ty21a model that constitutively expresses the GtgE protease from S. typhimurium, allowing this vector to be used in animal trials, thus enhancing the likelihood of favorable results that could quickly transition to clinical trials. From the current perspective, it is necessary to explore a greater diversity of bacterial vectors and find the best combination of implemented attenuations, generating personalized models that guarantee the maximum effectiveness in cancer therapy and vaccination.
Keywords: bacterial based vaccination; bacterial cancer; bactofection; gene therapy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Increasing the bactofection capacity of a mammalian expression vector by removal of the f1 ori.Cancer Gene Ther. 2019 Jul;26(7-8):183-194. doi: 10.1038/s41417-018-0039-9. Epub 2018 Aug 13. Cancer Gene Ther. 2019. PMID: 30100607 Free PMC article.
-
Bacterial Therapy of Cancer: Promises, Limitations, and Insights for Future Directions.Front Microbiol. 2018 Jan 23;9:16. doi: 10.3389/fmicb.2018.00016. eCollection 2018. Front Microbiol. 2018. PMID: 29472896 Free PMC article.
-
Bacteria in gene therapy: bactofection versus alternative gene therapy.Gene Ther. 2006 Jan;13(2):101-5. doi: 10.1038/sj.gt.3302635. Gene Ther. 2006. PMID: 16163379 Review.
-
Improving E. coli Bactofection by Expression of Bacteriophage ΦX174 Gene E.Methods Mol Biol. 2021;2211:3-14. doi: 10.1007/978-1-0716-0943-9_1. Methods Mol Biol. 2021. PMID: 33336266
-
Genetically engineered Salmonella Typhimurium: Recent advances in cancer therapy.Cancer Lett. 2019 Apr 28;448:168-181. doi: 10.1016/j.canlet.2019.01.037. Epub 2019 Feb 10. Cancer Lett. 2019. PMID: 30753837 Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

