Novel Administration Routes, Delivery Vectors, and Application of Vaccines Based on Biotechnologies: A Review
- PMID: 39340032
- PMCID: PMC11436249
- DOI: 10.3390/vaccines12091002
Novel Administration Routes, Delivery Vectors, and Application of Vaccines Based on Biotechnologies: A Review
Abstract
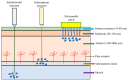
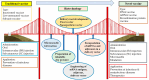
Traditional vaccines can be classified into inactivated vaccines, live attenuated vaccines, and subunit vaccines given orally or via intramuscular (IM) injection or subcutaneous (SC) injection for the prevention of infectious diseases. Recently, recombinant protein vaccines, DNA vaccines, mRNA vaccines, and multiple/alternative administering route vaccines (e.g., microneedle or inhalation) have been developed to make vaccines more secure, effective, tolerable, and universal for the public. In addition to preventing infectious diseases, novel vaccines have currently been developed or are being developed to prevent or cure noninfectious diseases, including cancer. These vaccine platforms have been developed using various biotechnologies such as viral vectors, nanoparticles, mRNA, recombination DNA, subunit, novel adjuvants, and other vaccine delivery systems. In this review, we will explore the development of novel vaccines applying biotechnologies, such as vaccines based on novel administration routes, vaccines based on novel vectors, including viruses and nanoparticles, vaccines applied for cancer prevention, and therapeutic vaccines.
Keywords: adjuvant; biotechnology; delivery system; mRNA; nanoparticle; recombination DNA; therapeutics; vaccine; vector.
Conflict of interest statement
The authors declare no conflicts of interests.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources