Efficacy and safety of thalidomide for recurrent aphthous stomatitis: a systematic review and meta-analysis of randomized controlled trials
- PMID: 39342210
- PMCID: PMC11438041
- DOI: 10.1186/s12903-024-04923-1
Efficacy and safety of thalidomide for recurrent aphthous stomatitis: a systematic review and meta-analysis of randomized controlled trials
Abstract
Background: Recurrent aphthous stomatitis (RAS) is considered as the most common oral mucosal lesion affecting up to 25% of people worldwide. Thalidomide has been reported for the treatment of RAS, but the evidence has not been systematically evaluated. We first systematically reviewed the efficacy and safety of thalidomide for the treatment of RAS.
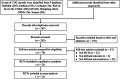
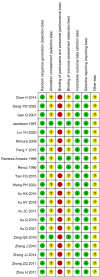
Methods: We searched The Cochrane Library, PubMed, Scopus, Web of Science, Embase, China National Knowledge Infrastructure (CNKI), Chinese BioMedical Literature Database (CBM), Wanfang Data, and VIP information from inception to December 2023. Randomized controlled trials (RCTs) comparing thalidomide with control for RAS were included in the analysis. The primary outcome were complete response and overall response, and the secondary outcome were recurrence interval (RI), ulcer number and size, healing time, visual analogue scale (VAS), immunological data, and adverse events. Meta-analysis was conducted using the Review Manager 5.4 software.
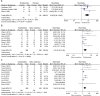
Results: Twenty-one trials involving 1668 patients were included in this review. The results of our meta-analysis showed that thalidomide significantly improved the complete response rate and overall response rate, prolonged the recurrence interval, accelerated the healing process, reduced the number and size of ulcers, and lowered TNF-α levels in the treatment of RAS. However, thalidomide significantly increases adverse events.
Results: Thalidomide has a significant benefit in the treatment of RAS. However, considering the potential side effects of thalidomide, it may be an optimal treatment option for major RAS patients or cases that do not respond to topical agents.
Trial registration: PROSPERO registration number: CRD42024495038.
Keywords: Meta-analysis; Recurrent aphthous stomatitis; Stomatitis, Aphthous; Systematic review; Thalidomide.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

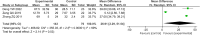
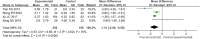

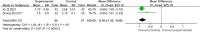
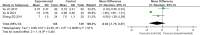

References
-
- Jonathan AS, Arbor DMD, Mich A. Recurrent aphthous stomatitis: an update. Oral Surg Oral Med Oral Pathol Oral Radiol. 1996;81(2):141–7. - PubMed
-
- Barrons RW. Treatment strategies for recurrent oral aphthous ulcers. Am J Health-Syst Pharm. 2001;58(1):41–50. - PubMed
-
- Wang ZH, Cao HB, Xiong JQ, Lu YL, Deng YX, Nan H, et al. Recent advances in the aetiology of recurrent aphthous stomatitis (RAS). Postgrad Med J. 2022;98(1155):57–66. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical

