Exercise training upregulates CD55 to suppress complement-mediated synaptic phagocytosis in Parkinson's disease
- PMID: 39342308
- PMCID: PMC11439226
- DOI: 10.1186/s12974-024-03234-0
Exercise training upregulates CD55 to suppress complement-mediated synaptic phagocytosis in Parkinson's disease
Abstract
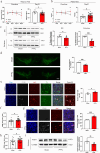
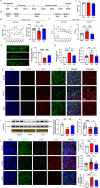
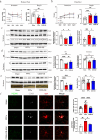
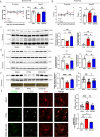
The primary pathological change in Parkinson's disease (PD) is the progressive degeneration of dopaminergic neurons in the substantia nigra. Additionally, excessive microglial activation and synaptic loss are also typical features observed in PD samples. Exercise trainings have been proven to improve PD symptoms, delay the disease progression as well as affect excessive microglial synaptic phagocytosis. In this study, we established a mouse model of PD by injecting mouse-derived α-synuclein preformed fibrils (M-α-syn PFFs) into the substantia nigra, and demonstrated that treadmill exercise inhibits microglial activation and synaptic phagocytosis in striatum. Using RNA-Seq and proteomics, we also found that PD involves excessive activation of the complement pathway which is closely related to over-activation of microglia and abnormal synaptic function. More importantly, exercise training can inhibit complement levels and complement-mediated microglial phagocytosis of synapses. It is probably triggered by CD55, as we observed that CD55 in the striatum significantly increased after exercise training and up-regulation of that molecule rescued motor deficits of PD mice, accompanied with reduced microglial synaptic phagocytosis in the striatum. This research elucidated the interplay among microglia, complement, and synapses, and analyzed the effects of exercise training on these factors. Our work also suggested CD55 as a complement-relevant candidate molecule for developing therapeutic strategies of PD.
Keywords: CD55; Complement pathway; Microglia; Parkinson’s disease; Proteomics and RNA-seq analysis; Synapses.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

