Ultrasound-Guided Percutaneous Biopsy With Needle Track Plugging in Patients With Focal Liver Lesions on an Outpatient Basis: A Randomized Controlled Trial
- PMID: 39344547
- PMCID: PMC11444846
- DOI: 10.3348/kjr.2024.0536
Ultrasound-Guided Percutaneous Biopsy With Needle Track Plugging in Patients With Focal Liver Lesions on an Outpatient Basis: A Randomized Controlled Trial
Abstract
Objective: The increasing utilization of various molecular tests for diagnosing and selecting treatments for patients with malignancies has led to a rising trend in both the frequency of biopsies and the required tissue volume. We aimed to compare the safety of outpatient ultrasound (US)-guided percutaneous liver biopsy (PLB) between the coaxial method with needle track plugging (NTP) and the conventional method.
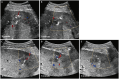
Materials and methods: This single-center, prospective, randomized controlled study was conducted from October 2022 to May 2023. Patients referred for US-guided PLB with target liver lesions measuring ≥1 cm and requiring ≥3 tissue cores were enrolled. Patients with severe coagulopathy or a substantial volume of ascites were excluded. Patients were randomly assigned to undergo PLB using either the coaxial method with NTP or the conventional method, in a 1:1 ratio, and were subsequently discharged after 2 hours. The primary endpoint was the presence of a patent track sign, defined as a linear color flow along the biopsy track on Doppler US, as an indication of bleeding. The secondary endpoints included clinically significant bleeding, delayed bleeding after discharge, and diagnostic yield. The incidences of these endpoints were compared between the two methods.
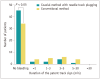
Results: A total of 107 patients completed the study protocol. Patent track signs were observed significantly less frequently in the coaxial method with NTP group than in the conventional method group: 16.7% (9/54) vs. 35.8% (19/53; P = 0.042). Clinically significant bleeding and delayed bleeding did not occur in either group, and both methods achieved a high diagnostic yield: 94.4% (51/54) vs. 98.1% (52/53; P = 0.624).
Conclusion: Compared with the conventional method, the coaxial method with NTP may potentially be safer, with a reduced risk of overall bleeding complications after PLB when retrieving ≥3 tissue cores. The coaxial method with NTP could be considered a viable option for acquiring multiple liver tissues on an outpatient basis.
Keywords: Bleeding; Hemorrhage; Image-guided biopsy; Liver; Prospective studies; Ultrasonography, Doppler.
Copyright © 2024 The Korean Society of Radiology.
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures
References
-
- Potretzke TA, Saling LJ, Middleton WD, Robinson KA. Bleeding complications after percutaneous liver biopsy: do subcapsular lesions pose a higher risk? AJR Am J Roentgenol. 2018;211:204–210. - PubMed
-
- Chi H, Hansen BE, Tang WY, Schouten JN, Sprengers D, Taimr P, et al. Multiple biopsy passes and the risk of complications of percutaneous liver biopsy. Eur J Gastroenterol Hepatol. 2017;29:36–41. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

