Carotid Baroreceptor Stimulation Ameliorates Pulmonary Arterial Remodeling in Rats With Hypoxia-Induced Pulmonary Hypertension
- PMID: 39344593
- PMCID: PMC11681457
- DOI: 10.1161/JAHA.124.035868
Carotid Baroreceptor Stimulation Ameliorates Pulmonary Arterial Remodeling in Rats With Hypoxia-Induced Pulmonary Hypertension
Abstract
Background: Sympathetic hyperactivity plays an important role in the initiation and maintenance of pulmonary hypertension. Carotid baroreceptor stimulation (CBS) is an effective autonomic neuromodulation therapy. We aim to investigate the effects of CBS on hypoxia-induced pulmonary hypertension and its underlying mechanisms.
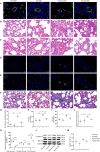
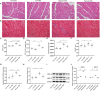
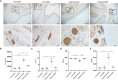
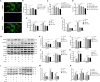
Methods and results: Rats were randomly assigned into 4 groups, including a Control-sham group (n=7), a Control-CBS group (n=7), a Hypoxia-sham group (n=10) and a Hypoxia-CBS group (n=10). Echocardiography, ECG, and hemodynamics examination were performed. Samples of blood, lung tissue, pulmonary arteries, and right ventricle were collected for the further analysis. In the in vivo study, CBS reduced wall thickness and muscularization degree in pulmonary arterioles, thereby improving pulmonary hemodynamics. Right ventricle hypertrophy, fibrosis and dysfunction were all improved. CBS rebalanced autonomic tone and reduced the density of sympathetic nerves around pulmonary artery trunks and bifurcations. RNA-seq analysis identified BDNF and periostin (POSTN) as key genes involved in hypoxia-induced pulmonary hypertension, and CBS downregulated the mRNA expression of BDNF and POSTN in rat pulmonary arteries. In the in vitro study, norepinephrine was found to promote pulmonary artery smooth muscle cell proliferation while upregulating BDNF and POSTN expression. The proliferative effect was alleviated by silence BDNF or POSTN.
Conclusions: Our results showed that CBS could rebalance autonomic tone, inhibit pulmonary arterial remodeling, and improve pulmonary hemodynamics and right ventricle function, thus delaying hypoxia-induced pulmonary hypertension progression. There may be a reciprocal interaction between POSTN and BDNF that is responsible for the underlying mechanism.
Keywords: POSTN; carotid baroreceptor stimulation; hypoxia; pulmonary hypertension; sympathetic tone.
Figures



Similar articles
-
Bioactive fraction of Rhodiola algida against chronic hypoxia-induced pulmonary arterial hypertension and its anti-proliferation mechanism in rats.J Ethnopharmacol. 2018 Apr 24;216:175-183. doi: 10.1016/j.jep.2018.01.010. Epub 2018 Jan 8. J Ethnopharmacol. 2018. PMID: 29325918
-
Reoxygenation Reverses Hypoxic Pulmonary Arterial Remodeling by Inducing Smooth Muscle Cell Apoptosis via Reactive Oxygen Species-Mediated Mitochondrial Dysfunction.J Am Heart Assoc. 2017 Jun 23;6(6):e005602. doi: 10.1161/JAHA.117.005602. J Am Heart Assoc. 2017. PMID: 28645933 Free PMC article.
-
Involvement of calcium-sensing receptors in hypoxia-induced vascular remodeling and pulmonary hypertension by promoting phenotypic modulation of small pulmonary arteries.Mol Cell Biochem. 2014 Nov;396(1-2):87-98. doi: 10.1007/s11010-014-2145-9. Epub 2014 Jul 26. Mol Cell Biochem. 2014. PMID: 25063217
-
Tsantan Sumtang Alleviates Chronic Hypoxia-Induced Pulmonary Hypertension by Inhibiting Proliferation of Pulmonary Vascular Cells.Biomed Res Int. 2018 Nov 28;2018:9504158. doi: 10.1155/2018/9504158. eCollection 2018. Biomed Res Int. 2018. PMID: 30622966 Free PMC article.
-
Astragaloside IV attenuates hypoxia‑induced pulmonary vascular remodeling via the Notch signaling pathway.Mol Med Rep. 2021 Jan;23(1):89. doi: 10.3892/mmr.2020.11726. Epub 2020 Nov 25. Mol Med Rep. 2021. PMID: 33236156 Free PMC article.
Cited by
-
Carotid baroreceptor stimulation attenuates obesity-related hypertension through sympathetic-driven IL- 22 restoration of intestinal homeostasis.Eur J Med Res. 2025 Apr 15;30(1):291. doi: 10.1186/s40001-025-02528-0. Eur J Med Res. 2025. PMID: 40234921 Free PMC article.
-
Pulmonary artery wall thickness and systemic sclerosis: influence of inflammation on vascular changes.Turk J Med Sci. 2025 Mar 24;55(3):644-651. doi: 10.55730/1300-0144.6011. eCollection 2025. Turk J Med Sci. 2025. PMID: 40686711 Free PMC article.
References
-
- Zelt JGE, Sugarman J, Weatherald J, Partridge ACR, Liang JC, Swiston J, Brunner N, Chandy G, Stewart DJ, Contreras‐Dominguez V, et al. Mortality trends in pulmonary arterial hypertension in Canada: a temporal analysis of survival per ESC/ERS guideline era. Eur Respir J. 2022;59:2101552. doi: 10.1183/13993003.01552-2021 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

