200 Years of The Haloform Reaction: Methods and Applications
- PMID: 39345024
- PMCID: PMC11653256
- DOI: 10.1002/chem.202403045
200 Years of The Haloform Reaction: Methods and Applications
Abstract
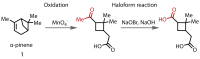
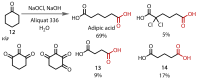
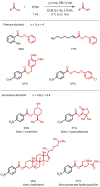
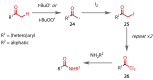
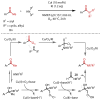
Discovered in 1822, the haloform reaction is one of the oldest synthetic organic reactions. The haloform reaction enables the synthesis of carboxylic acids, esters or amides from methyl ketones. The reaction proceeds via exhaustive α-halogenation and then substitution by a nucleophile to liberate a haloform. The methyl group therefore behaves as a masked leaving group. The reaction methodology has undergone several important developments in the last 200 years, transitioning from a diagnostic test of methyl ketones to a synthetically useful tool for accessing complex esters and amides. The success of the general approach has been exhibited through the use of the reaction in the synthesis of many different complex molecules in fields ranging from natural product synthesis, pharmaceuticals, agrochemicals, fragrants and flavourings. The reaction has not been extensively reviewed since 1934. Therefore, herein we provide details of the history and mechanism of the haloform reaction, as well as an overview of the developments in the methodology and a survey of examples, particularly in natural product synthesis, in which the haloform reaction has been used.
Keywords: Carboxylic acid; Ester; Haloform; Halogenation; Oxidation.
© 2024 The Author(s). Chemistry - A European Journal published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
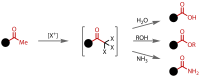



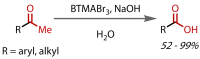
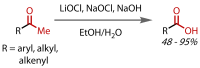
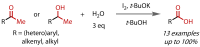
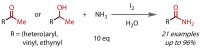

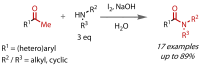
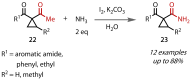
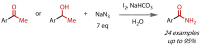
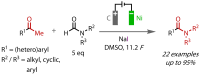

Similar articles
-
A Stoichiometric Haloform Coupling for Ester Synthesis with Secondary Alcohols.Angew Chem Int Ed Engl. 2024 May 21;63(21):e202400570. doi: 10.1002/anie.202400570. Epub 2024 Apr 18. Angew Chem Int Ed Engl. 2024. PMID: 38533790 Free PMC article.
-
Synthesis of 13 C-labeled parabens from isotopically enriched phenols using the Houben-Hoesch reaction.J Labelled Comp Radiopharm. 2022 Jul;65(9):254-263. doi: 10.1002/jlcr.3992. Epub 2022 Jul 26. J Labelled Comp Radiopharm. 2022. PMID: 35868986
-
Divergent Transformations of Aromatic Esters: Decarbonylative Coupling, Ester Dance, Aryl Exchange, and Deoxygenative Coupling.Acc Chem Res. 2024 Jun 18;57(12):1747-1760. doi: 10.1021/acs.accounts.4c00233. Epub 2024 May 31. Acc Chem Res. 2024. PMID: 38819671 Free PMC article.
-
Recent methods for the synthesis of α-acyloxy ketones.Org Biomol Chem. 2018 Dec 12;16(48):9334-9348. doi: 10.1039/c8ob02881h. Org Biomol Chem. 2018. PMID: 30516787 Review.
-
Visible-Light-Induced Catalytic Selective Halogenation with Photocatalyst.Molecules. 2021 Dec 5;26(23):7380. doi: 10.3390/molecules26237380. Molecules. 2021. PMID: 34885962 Free PMC article. Review.
References
-
- Fuson R. C., Bull B. A., Chem. Rev. 1934, 15, 275–309.
-
- VanArendonk A. M., Cupery M. E., J. Am. Chem. Soc. 1931, 53, 3184–3186.
-
- Minisci F., Recupero F., Fontana F., Bjørsvik H.-R., Liguori L., Synlett 2002, 2002, 0610–0612.
-
- Chiba S., Zhang L., Ang G. Y., Hui B. W.-Q., Org. Lett. 2010, 12, 2052–2055. - PubMed
-
- Kawata A., Takata K., Kuninobu Y., Takai K., Angew. Chemie Int. Ed. 2007, 46, 7793–7795. - PubMed
Publication types
Grants and funding
- University Research Fellowship and Enhancement awards/Royal Society
- 949821, SENF/HORIZON EUROPE European Research Council
- EP/S024107/1/EPSRC Centre for Doctoral Training in Technology Enhanced Chemical Synthesis
- EP/T001631/1/Engineering and Physical Sciences Research Council
- EP/L016443/1/Engineering and Physical Sciences Research Council
LinkOut - more resources
Full Text Sources

