The leukotriene receptor antagonist montelukast as a potential therapeutic adjuvant in multiple sclerosis - a review
- PMID: 39346564
- PMCID: PMC11427386
- DOI: 10.3389/fphar.2024.1450493
The leukotriene receptor antagonist montelukast as a potential therapeutic adjuvant in multiple sclerosis - a review
Abstract
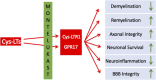
Multiple Sclerosis (MS) is a multifactorial autoimmune disease of the central nervous system (CNS). It is characterized by a heightened activation of the immune system with ensuing inflammation, demyelination and neurodegeneration with consequences such as motor, sensory, cognitive, as well as autonomic dysfunctions. While a range of immune-modulatory drugs have shown certain efficacy in alleviating pathology and symptoms, none of the currently available therapeutics regenerates the damaged CNS to restore function. There is emerging evidence for leukotrienes and leukotriene receptors being involved in the various aspects of the MS pathology including neuroinflammation and de/remyelination. Moreover, leukotriene receptor antagonists such as the asthma drug montelukast diminish inflammation and promote regeneration/remyelination. Indeed, montelukast has successfully been tested in animal models of MS and a recent retrospective case-control study suggests that montelukast treatment reduces relapses in patients with MS. Therefore, we propose montelukast as a therapeutic adjuvant to the standard immune-modulatory drugs with the potential to reduce pathology and promote structural and functional restoration. Here, we review the current knowledge on MS, its pathology, and on the potential of leukotriene receptor antagonists as therapeutics for MS.
Keywords: drug development; neuroinflammation; regeneration; remyelination; restoration.
Copyright © 2024 Pietrantonio, Serreqi, Zerbe, Svenningsson and Aigner.
Conflict of interest statement
FP, AS, and HZ are employees at IntelgenX corp., which holds patents in the use of montelukast in neurodegenerative diseases. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision
Figures
Similar articles
-
Montelukast as a repurposable additive drug for standard-efficacy multiple sclerosis treatment: Emulating clinical trials with retrospective administrative health claims data.Mult Scler. 2024 May;30(6):696-706. doi: 10.1177/13524585241240398. Epub 2024 Apr 25. Mult Scler. 2024. PMID: 38660773 Free PMC article.
-
Montelukast alleviates inflammation in experimental autoimmune encephalomyelitis by altering Th17 differentiation in a mouse model.Immunology. 2021 Jun;163(2):185-200. doi: 10.1111/imm.13308. Epub 2021 Feb 28. Immunology. 2021. PMID: 33480040 Free PMC article.
-
Animal models to investigate the effects of inflammation on remyelination in multiple sclerosis.Front Mol Neurosci. 2022 Nov 3;15:995477. doi: 10.3389/fnmol.2022.995477. eCollection 2022. Front Mol Neurosci. 2022. PMID: 36407761 Free PMC article. Review.
-
Neuronal injury in chronic CNS inflammation.Best Pract Res Clin Anaesthesiol. 2010 Dec;24(4):551-62. doi: 10.1016/j.bpa.2010.11.001. Epub 2010 Nov 29. Best Pract Res Clin Anaesthesiol. 2010. PMID: 21619866 Review.
-
Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years.Pediatrics. 2001 Sep;108(3):E48. doi: 10.1542/peds.108.3.e48. Pediatrics. 2001. PMID: 11533366 Clinical Trial.
Cited by
-
The Role of IL-23 in the Development of Inflammatory Diseases.Biology (Basel). 2025 Mar 27;14(4):347. doi: 10.3390/biology14040347. Biology (Basel). 2025. PMID: 40282212 Free PMC article. Review.
References
-
- Bäck M., Dahlén S. E., Drazen J. M., Evans J. F., Serhan C. N., Shimizu T., et al. (2011). International Union of Basic and Clinical Pharmacology. LXXXIV: leukotriene receptor nomenclature, distribution, and pathophysiological functions. Pharmacol. Rev. 63 (3), 539–584. 10.1124/pr.110.004184 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources