Tamsulosin and Dutasteride Combination Therapy for Asian Men With Moderate-to-Severe Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia: A Systematic Review of Clinical Considerations That Influence the Prescription
- PMID: 39346571
- PMCID: PMC11426169
- DOI: 10.14740/jocmr5255
Tamsulosin and Dutasteride Combination Therapy for Asian Men With Moderate-to-Severe Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia: A Systematic Review of Clinical Considerations That Influence the Prescription
Abstract
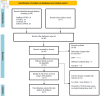
The goal of combination therapy for moderate-to-severe lower urinary tract symptoms secondary to benign prostatic hyperplasia (LUTS/BPH) is to ease both the dynamic and static symptoms by using agents that have complementary mechanisms of action. Similar to prescribing other drugs, LUTS/BPH combination therapy has been affected by multiple factors. Previous qualitative research discussed the individual perspectives that influenced combination therapy administration. Yet, until recently, there has been limited interest in clinical reasons that physicians have to consider before prescribing LUTS/BPH combination treatment. This systematic review aimed to identify the clinical considerations that influence the decision to prescribe combination therapy of tamsulosin 0.4 mg + dutasteride 0.5 mg for Asian men with LUTS/BPH. This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A systematic search was performed in databases Medline, CINAHL, the Cochrane Library, and Embase from inception until January 2024 using Medical Subject Headings (MeSH) terms and keywords with truncation for alternative acronyms. A citation search was performed to gather works of literature on LUTS/BPH combination treatment in addition to the "PICO" framework for search terms. Five English-language primary randomized controlled trials (RCTs) were included in the narrative analysis using the Critical Appraisal Skills Program (CASP) checklist after critical appraisal. Several dosages of tamsulosin (0.2 mg and 0.4 mg) have been administered in LUTS/BPH combination treatment over the last few decades despite 0.2 mg tamsulosin being standardized as an effective regime in Asian countries. A remarkable correlation between prostate volume (PV) and prostate-specific antigen (PSA) was found in Asian men, which requires higher PSA secretion to enlarge each prostate unit and causes an increased risk of moderate-to-severe LUTS. Additionally, BPH baseline variables may lead to a different response to combination therapy, especially the PV and PSA differences. In conclusion, compared with Caucasian men, a significantly higher risk of moderate-to-severe LUTS was found in Asian men. Initiation of combination therapy, especially dutasteride, depends on a larger PV (≥ 30 mL); it is possible, therefore, that earlier PV and PSA examinations and baseline variables assessments ought to be performed by physicians before the combination therapy prescription. Alternative treatment options may be considered for a patient who prefers an active pattern of sexual activity during their BPH combined pharmacotherapy. These clinical considerations may influence the prescription of tamsulosin 0.4 mg + dutasteride 0.5 mg combination therapy for Asian men with moderate-to-severe LUTS/BPH. This study was registered on PROSPERO (CRD42024575528).
Keywords: Benign prostatic hyperplasia; Clinical consideration; Combination therapy; Lower urinary tract symptoms; Prescription; Systematic review.
Copyright 2024, Yang et al.
Conflict of interest statement
The authors declare that they do not have a conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous