Synthesis and In Silico Evaluation of Piperazine-Substituted 2,3-Dichloro-5,8-dihydroxy-1,4-naphthoquinone Derivatives as Potential PARP-1 Inhibitors
- PMID: 39346823
- PMCID: PMC11425603
- DOI: 10.1021/acsomega.4c04915
Synthesis and In Silico Evaluation of Piperazine-Substituted 2,3-Dichloro-5,8-dihydroxy-1,4-naphthoquinone Derivatives as Potential PARP-1 Inhibitors
Abstract
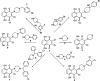
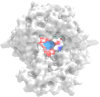
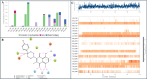
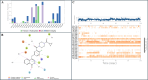
PARP-1 (poly(ADP-ribose)-polymerase 1) inhibitors are vital in synthetic lethality, primarily due to their specificity for PARP-1 over PARP-2 (PARP-1 > PARP-2). This specificity is crucial as it allows precise inhibition of PARP-1 in tumor cells with Breast Cancer 1 protein (BRCA1) or BRCA2 deficiencies. The development of highly specific PARP-1 inhibitors not only meets the therapeutic needs of tumor treatment but also has the potential to minimize the adverse effects associated with nonselective PARP-2 inhibition. In this study, a series of novel 2,3-dichloro-5,8-dihydroxy-1,4-naphthoquinone (DDNO) derivatives were synthesized, characterized, and evaluated regarding their PARP-1 inhibitory and cytotoxic activity. Compound 3 exhibited the highest cytotoxic potential against all cell lines, except for MDA-MB-231 cells. The inhibitory potential of these molecules against PARP-1 was evaluated through in silico molecular docking and molecular dynamics studies. Notably, compounds 5, 9, and 13 exhibited significant inhibitory activity in silico results, interacting with critical amino acids known to be important for PARP-1 inhibition during simulations. These compounds exhibited target-specific and strong binding profiles, with docking scores of -7.17, -7.41, and -7.37 kcal/mol, respectively, and MM/GBSA scores of -52.51, -43.77, and -62.87 kcal/mol, respectively. These novel compounds (DDNO derivatives) hold promise as potential PARP-1 inhibitors for the development of targeted therapeutics against cancer.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Synthesis of Some Benzimidazole-derived Molecules and their Effects on PARP-1 Activity and MDA-MB-231, MDA-MB-436, MDA-MB-468 Breast Cancer Cell Viability.Anticancer Agents Med Chem. 2020;20(14):1728-1738. doi: 10.2174/1871520620666200502001953. Anticancer Agents Med Chem. 2020. PMID: 32357823
-
In-silico method for elucidation of prodigiosin as PARP-1 inhibitor a prime target of Triple-negative breast cancer.Bioorg Chem. 2023 Sep;138:106618. doi: 10.1016/j.bioorg.2023.106618. Epub 2023 May 19. Bioorg Chem. 2023. PMID: 37244231
-
Design, synthesis, biological evaluation and molecular docking study of novel urea-based benzamide derivatives as potent poly(ADP-ribose) polymerase-1 (PARP-1) inhibitors.Eur J Med Chem. 2022 Dec 5;243:114790. doi: 10.1016/j.ejmech.2022.114790. Epub 2022 Sep 23. Eur J Med Chem. 2022. PMID: 36183505
-
PARP inhibitors in breast cancer: Bringing synthetic lethality to the bedside.Cancer. 2018 Jun 15;124(12):2498-2506. doi: 10.1002/cncr.31307. Epub 2018 Apr 16. Cancer. 2018. PMID: 29660759 Free PMC article. Review.
-
PARP (Poly ADP-Ribose Polymerase) inhibitors for locally advanced or metastatic breast cancer.Cochrane Database Syst Rev. 2021 Apr 22;4(4):CD011395. doi: 10.1002/14651858.CD011395.pub2. Cochrane Database Syst Rev. 2021. PMID: 33886122 Free PMC article.
References
-
- Kerns R. J.; Rybak M. J.; Kaatz G. W.; Vaka F.; Cha R.; Grucz R. G.; Diwadkar V. U. Structural Features of Piperazinyl-Linked Ciprofloxacin Dimers Required for Activity against Drug-Resistant Strains of Staphylococcus Aureus. Bioorg. Med. Chem. Lett. 2003, 13 (13), 2109–2112. 10.1016/S0960-894X(03)00376-7. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
