Single-arm trial of neoadjuvant ipilimumab plus nivolumab with chemoradiotherapy in patients with resectable and borderline resectable lung cancer: the INCREASE study
- PMID: 39349061
- PMCID: PMC11448277
- DOI: 10.1136/jitc-2024-009799
Single-arm trial of neoadjuvant ipilimumab plus nivolumab with chemoradiotherapy in patients with resectable and borderline resectable lung cancer: the INCREASE study
Abstract
Background: In non-small cell lung cancer (NSCLC), chemoradiotherapy (CRT) yields pathological complete response (pCR) rates of approximately 30%. We investigated using ipilimumab plus nivolumab (IPI-NIVO) with neoadjuvant CRT in resectable, and borderline resectable NSCLC.
Methods: This single-arm, phase-II trial enrolled operable T3-4N0-2 patients with NSCLC without oncogenic drivers. Primary study endpoints were safety, major pathological response (MPR) and pCR. Treatment encompassed platinum-doublet concurrent CRT, IPI 1 mg/kg intravenous and NIVO 360 mg intravenous on day-1, followed by chemotherapy plus NIVO 360 mg 3 weeks later. Thoracic radiotherapy was 50 or 60 Gy, in once-daily doses of 2 Gy. Resections were 6 weeks post-radiotherapy.
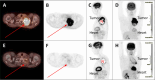
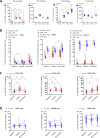
Results: In a total of 30 patients in the intention-to-treat (ITT) population, grades 3-4 treatment-related adverse events (TRAEs) occurred in 70%, one TRAE grade 5 late-onset pneumonitis on day 96 post-surgery (1/30, 3.3%) occurred, and one non-TRAE COVID-19 death (1/30, 3.3%). pCR and MPR were achieved in 50% (15/30) and 63% (19/30) of the ITT; and in 58% (15/26) and 73% (19/26) of the 26 patients who underwent surgery, respectively. Postoperative melanoma was seen in one non-pCR patient. The R0 rate was 100% (26/26), and no patient failed surgery due to TRAEs. In peripheral blood, proliferative CD8+ T cells were increased, while proliferative regulatory T cells (Tregs) were not. On-treatment, pCR-positives had higher CD8+CD39+ T cells and lower HLA-DR+ Tregs.
Conclusions: Neoadjuvant IPI-NIVO-CRT in T3-4N0-2 NSCLC showed acceptable safety with pCR and MPR in 58% and 73% of operated patients, respectively. No patient failed surgery due to TRAEs.
Trial registration number: NCT04245514.
Keywords: Chemotherapy; Ipilimumab; Nivolumab; Radiotherapy.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: The authors have disclosed the following conflicts of interest: DEO-L received grants from Janssen and Curium, honoraria from EANM, EAU, ESMO, and Curium, travel support from EANM, ESMO, EAU, and Bayer, and serves on a Bayer advisory board. CD received institutional fees for advisory roles with BMS, AstraZeneca, and MSD. SMSH has research contracts with several pharmaceutical companies and received honoraria from Janssen. IB’s institution received support from BMS, AstraZeneca, and Boehringer Ingelheim, and he received honoraria from AstraZeneca, MSD, and BMS. FLS received a research grant from ViewRay and honoraria from AstraZeneca. SS’s institution received grants from Varian, ViewRay, and AstraZeneca; he received consulting fees and honoraria from AstraZeneca and MSD, and serves on advisory boards for AstraZeneca and MSD. TDdG’s institution received grants from Idera Pharmaceuticals, consulting fees from LAVA Therapeutics and Mendus, and she holds stock in LAVA Therapeutics. All other authors have declared no conflicts of interest.
Figures


References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
