EZH2-dependent myelination following sciatic nerve injury
- PMID: 39359095
- PMCID: PMC11759024
- DOI: 10.4103/NRR.NRR-D-23-02040
EZH2-dependent myelination following sciatic nerve injury
Abstract
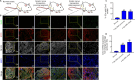
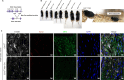
JOURNAL/nrgr/04.03/01300535-202508000-00028/figure1/v/2024-09-30T120553Z/r/image-tiff Demyelination and remyelination have been major focal points in the study of peripheral nerve regeneration following peripheral nerve injury. Notably, the gene regulatory network of regenerated myelin differs from that of native myelin. Silencing of enhancer of zeste homolog 2 (EZH2) hinders the differentiation, maturation, and myelination of Schwann cells in vitro. To further determine the role of EZH2 in myelination and recovery post-peripheral nerve injury, conditional knockout mice lacking Ezh2 in Schwann cells (Ezh2fl/fl;Dhh-Cre and Ezh2fl/fl;Mpz-Cre) were generated. Our results show that a significant proportion of axons in the sciatic nerve of Ezh2-depleted mice remain unmyelinated. This highlights the crucial role of Ezh2 in initiating Schwann cell myelination. Furthermore, we observed that 21 days after inducing a sciatic nerve crush injury in these mice, most axons had remyelinated at the injury site in the control nerve, while Ezh2fl/fl;Mpz-Cre mice had significantly fewer remyelinated axons compared with their wild-type littermates. This suggests that the absence of Ezh2 in Schwann cells impairs myelin formation and remyelination. In conclusion, EZH2 has emerged as a pivotal regulatory factor in the process of demyelination and myelin regeneration following peripheral nerve injury. Modulating EZH2 activity during these processes may offer a promising therapeutic target for the treatment of peripheral nerve injuries.
Copyright © 2025 Copyright: © 2025 Neural Regeneration Research.
Conflict of interest statement
Figures



References
-
- Bitgood MJ, McMahon AP. Hedgehog and Bmp genes are coexpressed at many diverse sites of cell-cell interaction in the mouse embryo. Dev Biol. 1995;172:126–138. - PubMed
LinkOut - more resources
Full Text Sources

